A Tetraploid Rhododendron carolinianum
August E. Kehr
Geneticist, Crops Research Division
Agricultural Research Service, U.S. Department of Agriculture
Plant Industry Station, Beltsville, Maryland
In the July 1966 issue of the American Rhododendron Society Bulletin, I described the larger, heavier textured flowers produced on plants as a result of doubling their chromosome numbers through colchicine techniques (1). At that time no examples were given for rhododendrons because no one had successfully doubled the chromosome numbers in this genus. The purpose of this article is to describe the development of a tetraploid form of the species Rhododendron carolinianum Rehd. through colchicine treatment of germinating seed. This is the first example of the induction of polyploid forms in broad-leaved species of rhododendrons. Colchicine-induced tetraploids in azaleas were developed at the Plant Industry Station, Beltsville, Maryland, in 1968, by treating stem buds with colchicine (2). This method has not been successful for me to date on broad-leaved rhododendrons. Consequently, I believe the seed treatment method is more effective with the broad-leaved types.
Methods and Materials
At least 1/2 ounce of seed of
R. carolinianum
was collected by Mr. George Miller, Hanover, Pennsylvania, in the fall of 1963, from plants of a white form growing in his yard. This seed was planted on May 9, 1964, in Petri-dishes on 10 percent water agar to which an aqueous solution of colchicine was added to make up a concentration of .0025 percent colchicine. The seed was germinated on the colchicine agar medium and planted on June 14 on milled sphagnum moss for further growth. The germinating seed and seedlings were kept under continuous fluorescent lights at a temperature of 85°F. It is probable than an absorbent paper saturated with a .0025 percent colchicine solution would work equally well as water agar as a medium for germinating seeds.
As the seedlings reached the two cotyledon stage, I used a tweezer to remove and discard the faster-growing ones, leaving the slower-growing seedlings to develop. Seedlings affected by the colchicine treatment tend to grow slower than the normal, unaffected seedlings. I estimate that at least 98 percent of the seedlings was removed at this stage, leaving only 2 percent to grow on. I repeated this thinning process at later intervals, to remove all fast growing seedlings. The remaining seedlings were smaller, and had slightly deeper green color, thicker stems, and fleshier cotyledons than the discarded seedlings. When the remaining seedlings reached the second true leaf stage, they were transplanted to flats. Between 20 and 30 plants were planted outside in the spring of 1965.
Results and Conclusions
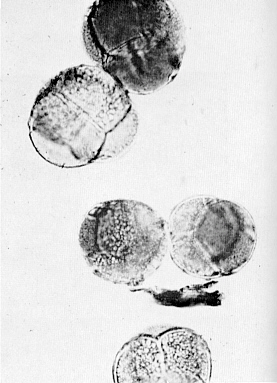
The first of this group flowered in the spring of 1969. The size and texture of the flowers indicated that the plant was a polyploid, and most probably a tetraploid. The flowers measured up to 21/2 inches in diameter (see Fig. 1) and were unusually heavy textured. The pale pink, almost white flowers were reflexed backwards, so that the flowers were reverse saucer shaped, in contrast to the tubular shape of the usual diploids.

|
|
Fig. 1. Flowers of tetraploid and three
diploid forms of R. carolinianum |
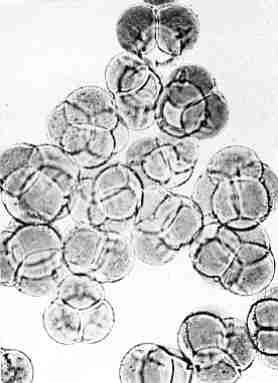
Preliminary chromosome counts made by Mr. Tom Miller, Hanover, Pennsylvania, indicate more than 26, the diploid number. Pollen size of the plant is uniformly larger than the pollen of a diploid taken as a check (see Fig. 2 and 3). Likewise the stomates were all larger than those of the diploid check plants. In general tetraploids can be expected to have pollen and stomate cells which have about 1-1/4 times the diameter of diploid cells.
|
|
|
Fig. 2. Pollen of Diploid
R. carolinianum (magnified 400x) Photo by L. C. Frazier |
Fig. 3. Pollen of Tetraploid
R. carolinianum (magnified 400x) Photo by L. C. Frazier |
Likewise the leaves of the plants in question are much broader than the average diploid leaves (see Fig. 4). The leaf length-width ratio for the tetraploid is 1:1.8, while the average for three nearby diploids is 1:2.0. The lower length-width ratio is a very common expression of a polyploid condition.

|
|
Fig. 4. Leaves of tetraploid
R. carolinianum
left, and diploids right.
Note the wider width of the tetraploid leaf as compared to its length. |
Based on evidence to date, such as the larger than normal stomates, pollen, flowers, and leaves, the plant appears to be tetraploid in the first 2 histogenic layers, L-I and L-II. An exact chromosome number of the root tips is required to ascertain the ploidy of L-III, the third or innermost histogenic layer. Mr. Miller's preliminary counts indicate that L-III is probably tetraploid. Thus it seems that the plant has tetraploid tissue in all 3 histogenic layers. Vegetative propagation or historical studies of the shoot meristem in the future should reveal whether or not this plant is cytochemical or completely tetraploid.
The plant is self fertile and selfed seed was obtained in the fall of 1969. The selfed seedlings likewise appear to be polyploids. This confirmed the polyploid nature of L-II revealed by large pollen grains.
A few seedlings were obtained when pollen from this plant was used on the Gable hybrid, 'Mincar', (
R. minus
Michaux x
R. carolinianum
), and when used as seed parent in a cross with
R. chrysodoron
Tagg ex Hutch.
Although no polyploid species have previously been reported in the Carolinianum series, polyploid species exist naturally in eight series of scaly rhododendrons (Table 1). It is interesting that the Lapponicum and Triflorum series have an especially high incidence of polyploid species reported. However, the highest chromosome numbers are found in the Maddenii and Heliolepis series where octoploid and duodeciploid (12 ploid) forms are found.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
1
From Darlington. C. D., and A. P. Wylie, 1956, Chromosome Atlas of Flowering
Plants, MacMillan Co., 519 pp.
2
Triploid found in
R. diaprepes
Balf. f. et W. W. Sm. (Gargantua).
3
In
R. manipurense
Balf. f. et Watt both 78 and 156 chromosome numbers were
reported. The 156 chromosome count is the highest reported in the genus.
4
R. pholidotum
Balf. f. et W. W. Sm.
It is likewise noteworthy that with the single exception of the triploid form found in R. diaprepes and given the cultivar name of 'Gargantua', there have been no polyploid species reported in the non-scaly rhododendrons. I am waiting for plants of R. maximum L. treated with colchicine in the manner described in this report, to flower, and I am hopeful that polyploid forms will be found.
Literature Cited
(1). Kehr, A. E., 1966, Breeding for a Purpose, Amer. Rhod. Soc. Bull. 20:131-141.
(2). Pryor, R. L., and L. C. Frazier, 1968, Colchicine-Induced Tetraploid Azaleas. Hort Science 3:283-284.