The Life Cycle of a Rhododendron
P. G. Valder, Professor of Botany, University of Sydney, Sydney, Australia
Notes accompanying an exhibit for the Pacific Rhododendron Society, October, 1970

|
|
FIG. 12. Flower of
R. macgregoriae
, partially
dissected to reveal its structure. 1-calyx; 2-corolla; 3-disc, which secretes nectar; 4-filament; 5-anther; 6-ovary; 7-style; 8-stigma. |
The life cycle as it occurs in nature. Many enthusiasts have carried a rhododendron through its complete life cycle from pollination, through seed setting, harvest, and germination, back to flowering. Since not everyone has a really clear idea of what has happened, let us start by taking a close look at that most complex of organs, the flower, the important parts of which are illustrated in Figure 12.
The flower, like the rest of the plant, is made up of a large number of cells, each of which contains a nucleus, which in turn contains a number of chromosomes on which occur the genes, which determine every characteristic exhibited by the plant. The chromosome compliment of each nucleus is made up of two sets, identical in appearance but differing in genetic make-up, one of which has come from the pollen-parent and one from the seed-parent.
In each nucleus an exact copy is made of every chromosome, so that, when the nucleus divides, the group of chromosomes in each new cell is an exact replica of that which was present in the original cell before it divided. Thus in every cell the complete genetic make-up of the plant is recorded on an identical set of chromosomes.
It is because of this that botanists have been able to take a single cell from a plant and produce from it a complete plant, identical with that from which it was taken. We do the same sort of thing on a much less sophisticated scale when we propagate plants vegetatively. Mercifully, biologists have not yet succeeded in doing the same with human cells, or we might all be competing with younger but otherwise identical versions of ourselves.
The number of chromosomes in each nucleus of a particular organism is known, not surprisingly, as its chromosome number. For instance our chromosome number is 46, made up of a set of 23 from each parent. Most rhododendrons have a chromosome number of 26, made up of a set of 13 from each parent.
Some rhododendrons have more than two sets of chromosomes. Those with 52 have four sets, each parent contributing two and those with 78 have six sets, each parent contributing three. Occasionally rhododendrons are found with 39 chromosomes, and it is thought that these plants arose from crosses between parents with 52 and 26 chromosomes respectively, the former contributing two sets and the latter one. Plants with three sets, or any other, odd number, are usually sterile, since the chromosomes cannot divide into two equal groups. This is the most common reason for sterility in hybrid rhododendrons arising from crosses between parents with different chromosome numbers.
Now in the flowers there are some special cells in the young anthers and ovules which undergo a special sort of cell division. In the anthers, each of these cells divides into four, but each of these four cells gets only one chromosome of each type, a single set of 13 in most rhododendrons. These cells are the pollen grains and they remain joined together in groups of four.
Likewise in the ovules there are also special cells which divide into four. Unlike the anthers in which there are lots of such cells, each ovule has only one and, after it divides into four, only one cell survives. This then divides to form a group of cells called the embryo sac. one of the cells of which becomes the egg cell with a single set of 13 chromosomes. For further development to take place there must next be fertilization, which can only take place if it is preceded by another event, pollination, the transfer of pollen from the anthers to the stigma.
Most rhododendrons are pollinated by insects or birds, which are attracted to the flower by their appearance and, in some cases, scent, their reward being a meal of pollen or nectar.

|
|
FIG. 13. Flower of
R. macgregoriae
showing
pollen being shed through two pores at the apex of each anther. Note that it clings together in a sticky mass, and thus it can be pulled out from the anthers by insects or birds which touch it. Note also that the stigma is not yet mature and is situated behind the anthers, where it is unlikely to be pollinated. |
The anthers of rhododendrons open by means of pores at their tips (Fig. 13) and it is from these that the pollen is shed. Amongst the pollen grains of rhododendrons are fine threads, which are entangled with one another and with the pollen grains. When an insect or bird touches the tip of the anther, it pulls the pollen out through the pores in a tangled mass and then may move off to another flower, perhaps on another plant, and some of the pollen may be wiped off onto the stigma.
Most rhododendrons have evolved devices which make it probable that cross-pollination will occur. In some the stigma protrudes from the bud before it opens to reveal the anthers. In others, the anthers mature and shed their pollen before the stigma is mature. In some of these the style remains shorter than the filaments of the anthers and elongates, pushing the stigma forward, after the pollen has been removed. In yet others the anthers bend back after a time, exposing the stigma, making it more probable, as in the previous case, that it will be touched by a bird or insect visiting the flower. The mature stigma secretes a sugary fluid which has two main functions. Firstly it traps the pollen grains and secondly, it provides them with moisture and food materials for their further development. Also, if experience with other plants is any guide, it will probably be found that it inhibits the further development of pollen grains of most plants other than rhododendrons.
|
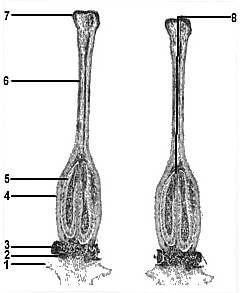
FIG. 14. Longitudinal section of the ovary, style, and stigma, showing the course followed by a pollen tube. Note that the cavities of the ovary contain large numbers of ovules. 1-calyx; 2-position of corolla attachment; 3-disc; 4-ovary; 5-ovules; 6-style; 7-stigma; 8-pollen grain. The fertilized egg nucleus commences dividing to form the embryo, each cell of which has 26 chromosomes, and the whole ovule gradually develops into the seed. |
After the rhododendron pollen grain has been deposited in this fluid it commences further development. Each pollen cell has a conspicuous pore in its wall and through this a tube develops which then grows into the tissue of the stigma, down the style, and into the ovule (Figure 14) where it discharges two nuclei into the embryo sac. One of these nuclei fuses with the nucleus of the egg cell to produce a cell with two sets (26) of chromosomes once more. This cell commences dividing and develops into the embryo of the next generation and the tissues surrounding it also develop further, the whole thing becoming the seed. Fertilization also triggers off the further development of the ovary which enlarges, as the seeds develop, becoming a fruit of the type known as a capsule.
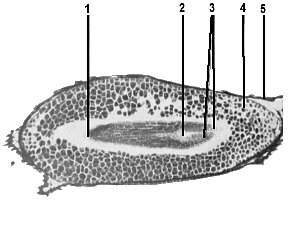
As the seed matures the embryo ceases development and becomes dormant. At this stage, however, there has already been considerable differentiation. The embryonic stem bears at its base an embryonic root, the radicle, and at its apex an embryonic growth bud, which will develop into the shoot (Figure 15). On either side of this are the two seed leaves, the cotyledons, and the whole embryo is surrounded by several layers of cells in which is stored sufficient food to enable the seedling to establish itself and commence manufacturing its own food.
|
FIG. 15. Longitudinal section of a mature seed, showing the seed coat surrounding the food storage tissue in which the embryo is embedded. 1-embryonic root; 2-embryonic shoot; 3-seed leaves (cotyledons); 4-food storage tissue; 5-seed coat |
The seeds are scattered when the capsules open and may be carried away by air currents or dispersed by other means. The seeds of many species have wing-like projections which assist aerial dispersal. The long-tailed seeds of the Malesian species are particularly well adapted for being blown away.
If the seed falls in a suitable place, it will germinate, sooner or later, when conditions of temperature and moisture are satisfactory. The seed absorbs moisture and the cells of the embryo commence dividing once more, the embryo continuing its development with the aid of the food materials stored in the tissue which surround it. A root emerges from the seed and grows down into the soil. The young stem grows up into the air and the seed-leaves are pulled from inside the seed-coat. The young seedling then becomes independent, its root system absorbing water and dissolved substances from the soil, and the shoot absorbing carbon dioxide and light energy. It then grows on and eventually flowers, the cycle being repeated.
Seed production ensures dispersal, continuity of the species in time, and variability, since each seedling has a different compliment of genes received from the pollen nucleus and egg cell from which it developed.
The development of the seedling is, of course, influenced by many environmental factors. Since gardeners are likely to provide adequate moisture and nutrients and some protection from diseases, pests and other adversities, the factors which will be considered here are day length and temperature.
In temperate climates most rhododendrons flower in the spring, grow in late spring and early summer, cease growth with their shoots terminating in flower or shoot-buds, ripen their seeds, and then become dormant prior to the onset of the cold weather. This dormancy is induced by a shortening of the days and is broken by subjection of the plant to a period of cold, followed by rising temperatures and increasing day-length. Thus the plant ensures that, in its natural habitat, it becomes dormant before winter arrives and doesn't flower and grow until the danger from frosts is over.
There is one feature of the behavior of rhododendron plants which seems worthy of mention. As happens with most plants, the roots of rhododendrons enter into complex relationships with fungi. The absorbing organs of the plants are thus not just roots, but roots invaded by a fungus. When such an association appears to benefit both partners we call it a symbiosis, and the symbiotic association between a root and a fungus is called a mycorrhiza.
As it happens, almost nothing is known concerning the type of mycorrhiza that occurs in rhododendrons, other members of the Ericaceae, members of the Epacridaceae, and certain other plants. The identity of the fungus or fungi has not been established nor has the function of the mycorrhiza been elucidated. It seems likely, however, that the position will be shown to be similar to that occurring in many other plants. For these it has been shown that, while the fungus is parasitic on the plant, the disadvantages of this are far outweighed by the increased efficiency of the mycorrhizal roots in taking up minerals, particularly phosphorus. It is probable that many plants growing under natural conditions in poor soils would be unable to survive were it not for these mycorrhizal associations. In cultivation, however, the plants will grow perfectly satisfactorily without the fungus, provided they are given adequate mineral nutrients.
The root systems of rhododendrons and their relatives are very characteristic in appearance. The main branches are rarely extensive and give rise to a mass of extremely fine and frequently branching rootlets. Some of the tropical epiphytic rhododendrons are described in the literature as having "thick, fleshy roots", a description which calls to mind the orchids. There is no evidence, however, that these are anything more than the main roots, which give rise to the fine rootlets in situations in which it is possible for them to develop. The fine roots grow through the soil as a result of the elongation brought about by the rapid division of a group of cells at the tip. These cells divide to produce the root cells on one side, and root-cap cells on the other. The root cap cells are loose and easily pushed off and, being continuously replenished, they protect the root tip from injury as it pushes through the soil.
In transverse section it can be seen that each rootlet has a single layer of large cells surrounding a fine central core of conducting tissue. The fungus grows around these roots and grows into the large outer cells and forms complex coils within them.

|
|
FIG. 16. Flower prepared for pollina-
tion, showing removal of the anthers and part of the corolla. This must be done before the bud opens as other- wise pollination may already have occurred. |
Some Practical Considerations For Hybridizers and Seedling Raisers.
For those interested in raising species or hybrids from seed, a detailed knowledge of the growth and reproduction of rhododendrons can be of considerable use. Some of the practical aspects are examined below.
Emasculation
Because of the strong tendency for cross-pollination to occur, open-pollinated seed is notoriously unreliable except from plants growing in the wild or which flower at a time when there is nothing else for them to be crossed with. Hence, under garden conditions hand pollination is essential, and even this must be combined with measures which render cross-pollination impossible.
Firstly, if a cross is to be attempted, all the anthers must be removed from the flowers of the seed parent before the buds open, preferably well before if you wish to avoid self-pollination, since in many species and hybrids the pollen is mature and can be easily dislodged several days before the flowers open. It is usually easiest to remove most of the corolla with scissors and then cut off the anthers. (Figure 16).
Pollination
Many authorities say that following the removal of the anthers, the flowers should then be covered with a bag and left till the stigmas mature before pollination is carried out. It is certainly easy to get the pollen to stick on if this is done, but it is much less bother to carry out the emasculation, pollination, bagging and labeling as part of the one operation. Because of the threads amongst the pollen grains a mass of pollen can usually be wound round the stigma even if it is not yet sticky. The proportion of "takes" following pollination at this time is no less than when it is carried out later when the stigmas have matured. If it proves too difficult to attach a mass of pollen to an immature stigma, it can be wetted with a drop of water, or even better, a 10% sugar solution (two ounces in one pint of water). Under laboratory conditions rhododendron pollen has been shown to germinate and grow very well in such a solution. However, with a little practice, it should not be necessary to have to resort to such devices.
|
|
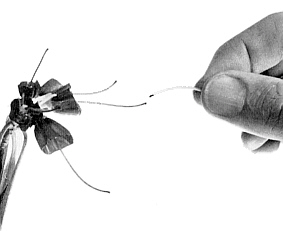
FIG. 17. Pollination is easily carried out using
an anther held by the filament. Anthers must be taken from unopened buds, since those from open flowers may be contaminated with pollen from elsewhere. |
The easiest way to pollinate a stigma is to hold an anther by the filament and touch the pollen against the stigma and allow it to be pulled through the pores (Figure 17). The use of camel-hair brushes, or forceps, is finicky and they have to be sterilized between each use if contamination with unwanted pollen is to be avoided. The hands, of course, should always be thoroughly washed if there is any chance of their carrying unwanted pollen. It should always be remembered that pollen should be taken from unopened flowers only, as otherwise one cannot be certain that it has not become mixed with pollen from other flowers.
Following pollination the flowers should be enclosed in a bag and left there till the stigmas wither and are no longer receptive. Brown paper bags are the best. They shade the flowers and allow movement of moisture and gases in and out. Plastic, cellophane, or waxed paper bags are much less satisfactory. They retain moisture, which encourages the development of moulds, and, if sunlight strikes them, the flowers heat up and may well be injured, since they are in a water saturated atmosphere and are not cooled by evaporation from their surfaces as they would otherwise be.
Failure to emasculate properly, to bag the flowers, and to take pollen only from unopened flowers are undoubtedly the factors responsible for some of the unlikely-sounding parentages recorded in the Rhododendron Stud Book.
Pollen Storage
Very often the most desirable crosses turn out to be those between species which do not flower at the same time, or in which there is no overlap between the opening of the last flowers of one and the first of the other. This problem can easily be overcome by storing pollen, which keeps quite well in a dry atmosphere, and will remain viable for quite long periods, probably several months, if kept in a refrigerator. It has recently been reported that dry pollen will keep for several years if frozen, and there is little doubt that pollen collected in the northern hemisphere in May could be used to pollinate flowers in the southern hemisphere in the following October.
If anthers are collected from flowers about to open and laid out in a matchbox or some other small container, and placed in the refrigerator with one end of the box slightly open, they will dry out and the pollen will remain viable for a couple of months at least, which is all most people want. The more sophisticated may wish to store their pollen in gelatin capsules over silica gel in a sealed container, although for most purposes this is an unnecessary refinement. However, it is quite easy to do. The anthers are placed in gelatin capsules, or folded in pieces of paper, which are just as good, and placed in a jar with some silica gel. The silica gel should be blue (if it is pink, heat it in the oven until it goes blue) and can be kept in place in the bottom of the jar with a layer of cotton wool. The jar should be tightly sealed to exclude moisture. If the gel starts to go pink it should be replaced or reheated at once.
The only alternative to pollen storage is a retardation or advancement of the flowering time of one or other of the proposed parents. This, of course, is much more bother and is unnecessary. Ordinarily it doesn't matter which of the parents is the seed parent as, in spite of the claims of some, geneticists are of the opinion that the progeny will be similar either way. However, if a cross is found not to take it is always worth trying the reverse cross.
Seed Harvesting
"In autumn, at the first sign of the capsules splitting, they should be carefully harvested and stored in a cool, dry place, etc., etc.,", say many authorities, and good advice it is too. Then when the capsules have dried out fully and split open, the seed can be shaken out, placed in a packet, and stored.
The impatient rhododendron breeder, however, may wish to be a bit more reckless. If the capsules are picked a month, or even more, before they are expected to mature and allowed to dry out, they can be broken open (they don't usually split if picked green) and viable seeds obtained. These usually look rather pale compared with those from mature capsules but, if they are sown at once, they germinate and a new generation gets under way earlier than would otherwise be possible.
It may be that these immature seeds do not keep as long as those from ripe capsules, but then they wouldn't have been harvested early if they weren't going to be sown at once.
Seed Storage
The same remarks apply here as did for pollen storage, except that seeds of most rhododendrons will keep for at least twelve months at room temperature. The seeds of the Malesian species and their hybrids seem to be short-lived, however, and don't seem to remain viable for long whatever you do. No doubt there has been no reason for the Malesian species to evolve a prolonged seed viability, since they come from tropical climates in which there is little variation the year round, whatever the altitude.
"The cool, dry place" we hear so much about is, of course, the best one in which to keep seeds of most kinds, since they stay alive longest at low temperature and humidity. In refrigerators most rhododendron seeds will remain viable for at least two years and perhaps much longer.
Seed Raising

|
|
FIG. 18. Pots sown with seeds are stood in
half an inch of water in a container, which is enclosed in a plastic bag. If the whole thing is stood in good light, but not direct sunlight, at 70- 75°F, germination should ensue within 14 days. |
As everyone knows, seeds need moisture, air, and warmth to germinate, and they won't get far after that if there isn't some light as well. An open compost with good water-retaining ability is the best and temperatures around 70-75°F., although most rhododendrons will germinate at somewhat lower temperatures. While everyone has his or her own favorite compost, pure peat seems very reliable and eucalyptus sawdust seems to work very well too. The surface should then be moistened and anchored down with a fine spray of water, such as that provided by an atomizer.
A simple way of raising seedlings is to plant the seeds in a small plastic pot, which is stood in half an inch of water in a tin or some other suitable container, and the whole thing enclosed in a plastic bag (Figure 18) and stood in good light but where no direct sunlight falls. When the cotyledons have expanded, it is a good idea to remove the bag and sprinkle the surface of the pot with sand (preferably sterilized by autoclaving or boiling in water) to anchor the seedlings. The bag should then be replaced but left open a little at one end. This reduces the humidity and the seedlings harden off. After about a week the bag can be removed permanently, then, a few days later, a little direct sunlight will do no harm.. The pot is kept damp by adding water to the tin as required.
If the seedlings are crowded, it is wise at this stage to thin them with forceps, so that those left are about a quarter of an inch apart. The pot can then be watered with a soluble fertilizer and subsequent applications made very three or four weeks. As soon as one or two leaves have expanded the pot can be removed from the tin and gentle overhead watering commenced.

|
|
FIG. 19. Provided temperatures are high
enough, rhododendrons can be kept growing continuously if kept near a light bulb which is left switched on. This prevents the onset of dormancy when the day length is short. The light bulb is used merely to prevent dormancy not to serve as a supplementary source of light for growth. |
It is usually a good idea to transplant the seedlings to individual pots before they get too crowded, as otherwise diseases, such as Botrytis blight, may break out. A watch should be kept too for mites and insect pests and appropriate control measures carried out where necessary.
Supplementary Light
Unless you live in a frost-free climate or have a glasshouse or growth room, it is not worth worrying about artificial light of any sort. However, if temperatures remain high enough for growth to take place throughout the year, supplementary light enables the seedlings to keep growing continuously. Otherwise, as the days shorten and temperatures begin to fall at the end of their first season, they will become dormant.
The response of plants to day-length is in fact very largely a response to night-length. As the nights lengthen the plant becomes dormant. By lighting the plants for a short period in the middle of the night, the dark period is divided into two short "nights" and no dormancy occurs. The plants just keep on growing, provided temperatures are high enough.
The intensity of light needed is very small, and, since the wavelengths required to affect the day-length response are at the red end of the spectrum, ordinary incandescent light bulbs are as good as anything. Without acquiring any complex apparatus, the desired effect can be achieved by hanging a bulb above the seedlings and leaving it on all night, or, more simply, all the time (Figure 19). The ordinary daylight provides the plants with the energy they need for growth and the light bulb stops them becoming dormant.
Sooner or later the plants will have to be transferred to the natural seasonal regime, and this should be done in late spring or summer so that the plants can adjust before the days shorten. Then they will become dormant in the usual manner in the autumn. However as a result of the early seed harvest and treatment with supplementary light, the seedlings should be pleasingly large 18 months after pollination.