Evolutionary Interactions Between Rhododendrons,
Pollinating Insects and Rust Fungi
Contributions from the Plant Genetics and Germplasm Institute
United States Department of Agriculture, Agricultural Research Center West
Beltsville, Maryland. Plant Introduction and Genetic Resources Investigation Paper No. 35
To be published in Annales of Botanicy Fennici
Elmer E. Leppik
Present address: Plant Genetics and Germplasm Institute
USDA, ARC-West, South Building, Room 326, Beltsville. Maryland
Development of rhododendrons from an ericoid stock presumably took place on the remnants of the former Laurasian super-continent in the Tertiary period. Extensive Tethys Sea between Laurasia and Gondwanaland in early Tertiary and desicated Mediterranean area during the Miocene, hindered the spread of these shrubs to the southern Continents.
Differentiation of rhododendron flowers from the gamopetalous ericoid type pursues the general trend of floral evolution from the pleomorphic and stereomorphic patterns toward bilateralism and zygomorphism, and from polymery toward pentamely.
Successive changes in floral patterns occurred mainly under the selective pressure of hymenopterous and lepitopterous pollinators. Bird pollination of rhododendrons is rare, so far reported only from Malaysia.
Heteroecious rhododendron rusts of the genus
Chrysomyxa
require
Picea
or
Tsuga
as alternate hosts, indicating the co-evolution of rhododendrons, spruces and hemlocks in a close neighborhood.
Additional key word,: evolution of rhododendron rusts,
Chrysoniyxa
, distribution of rhododendrons; distribution of rhododendron rusts.
I. Introduction
Two closely related and often united genera.
Rhododendron
and
Azalea
, are among the most delightful ornamentals frequently grown in homes, gardens. and parks. Their attractiveness is due mainly to the large-sized and brilliantly colored blossoms and to some extent to the elegant leaves and form of growth. If these ornamentals are properly - arranged in woody backgrounds, picturesque sceneries can be produced. While the rose is poetically called "Queen" of flowers. the rhododendron in its stately - majesty might well be entitled "King" (Bowers 1936, LEPPIK 1961).
In view of their natural variability and wide geographic distribution, rhododendrons and azaleas provide rich material for production of cultivars and hybrids of almost any color combination and growth form (BOWERS 1936, 1960, LEACH 1961). The large number of wild species and varieties, distributed from the Arctic to the Tropics, provides rich material for adaptation of these shrubs to various soils and altitudes in diverse climatic zones. Scientific exploration of this vast material enables us better evaluation of this natural source of beauty (KEHR 1972).
The intent of this study is to explore the fitness and adaptability of various foreign rhododendrons for the possible introduction to and acclimatization in the Fennoscandian region. Present geographic distribution, fossil evidence, pollination ecology, and specific rust flora of rhododendrons suggests the northern origin of these beautiful shrubs. Their modest, campanulate. monochromic flowers are also typical to northern areas. Yet there are no wild rhododendrons in northern Europe. except the
R. lapponicum
L., a dwarf arctic shrub of little ornamental value. Cultivation of rhododendrons in all Scandinavian countries, in spite of their beauty, is still relatively limited. But there are evidently neither decisive climatic nor edaphic factors that would hinder the growth of rhododendrons in northern Europe as first-class ornamentals.
Scanty fossil findings indicate that rhododendrons have been widespread in northern Europe during the Miocene and Pliocene epochs, 25-10 million years ago. Presumably these ancient plants were driven out from northern Europe or exterminated during the Pleistocene glaciation, about one million years ago. Post-glacial distribution of rhododendrons has been relatively slow and did not reach Europe, except in the Alps, Carpathian mountains and Arctic.

|
|
FIG18.
Rhododendron brachvcarpum
D.
Don ex G. Don ssp. tigerstedtii Nitz. Grown in the Botanical Garden Goteborg from seed brought by Dr. Tigerstedt from the Arboretum Mustila, Finland. Line drawing made by R. O. Hughes after photography of T. G. Nitzelius, Goteborg. |
Early trials with Korean and Japanese rhododendrons by Dr. Carl Gustav Tigerstedt in his world-known Mustila Arboretum in South Finland, demonstrated the winter hardiness and adaptibility of these beautiful shrubs to the hard climate of northern countries.
R. brachycarpum
Don ex G. Don introduced by Tigerstedt from the mountains of east-central Korea to Mustila, withstood -45° C (-49° F.) without any damage. In the year 1955 some samples of this and other Japanese and Korean rhododendrons from Mustila were planted in the Botanical Garden of Goteborg, south-west Sweden. D. T. G. NITZELIUS (1970, 1972) described one of these rhododendrons as
R. brachycarpum
ssp.
tigerstedtii
Nitz. ssp. nov. (Fig. 18). This writer has seen it in the Botanical Garden of Goteborg in September 13, 1971, as a strong, 4 m. high magnificent shrub. According to Nitzelius it blooms normally from middle June till August. This ssp, seems particularly fit for landscape ornamentation in the Fennoscandian region.
Detailed knowledge of the evolutionary history of rhododendrons is a necessary prerequisite of any breeding program that deals with production of new cultivars and hybrids. Some recent studies on floral evolution, (LEPPIK 1957, 1972), pollination ecology (FAEGRI and PIJL 1966) and rust relationship (LEPPIK 1973) provide a new approach to the origin and evolution of present-day rhododendrons.
II. Floral Characteristics and Evolutionary Differentiation of Rhododendron Flowers
Rhododendron flowers characteristically have a bell-shaped gamopetalous, penta-merous (seldom tetra-, or deca-) structure on the stereomorphic evolutionary level. Some common corolla types are campanulate, tubularcampanulate, funnelform, salverform, and urceolate (Fig. 19). There are usually twice as many stamens as corolla lobes, seldom the same number. Capsules are typically five celled, five valved, and many seeded. Flowers are commonly in terminal umbels, seldom in racemes (Fig. 20), or solitary.
|
|
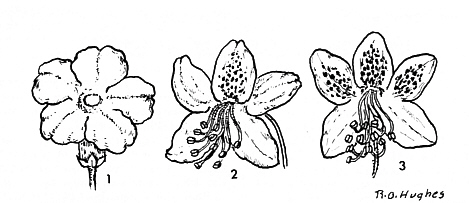
FIG19. Some Rhododendron flowers:
1. R. trichostomum var. ledoides Franch (= R. ledoides Balf. & Smith.) (Anthopogon ser.) radially symmetrical tubular corolla with pistil and stamens inside narrow corolla tube. Corolla pink. Plant introduced from Yunnan. 2. R. yunnanense Franch. (Tri-florum ser.). Bilateral pinkish corolla with red spotted upper petal and upward oriented pistil and stamens. 3. R. camtschaticum Pallas. (Camtschaticum ser.). Bilateral pinkish-purple corolla, three upper petals with red spots. (Drawn from photographs.) |
|
|
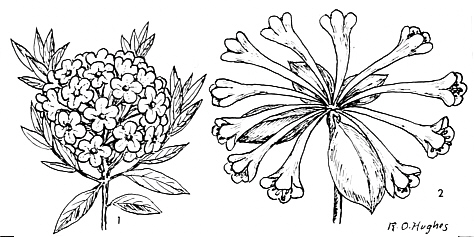
FIG.20. Some typical inflorescences of rhododendrons.
1. R. trichostomum var. ledoides Franch, flowers in a densely capitate, subglobose, terminal clusters. 2. R. carringtoniae F. v. Muell. with umbellate flowers. The long flower tube prevents short-tongued visitors from reaching the nectar. |
There are few exceptions to this general floral structure. Flowers of some series are considered to be more primitive than the majority of flowers in the genus, others more advanced. Thus the primitive Falconeri and Grande Series have 7-10 radially symmetrical corolla lobes, 12-18 stamens, and a 7-18 carpelled ovary. More advanced rhododendrons have pentamerous flowers (Fig. 21) with a clear tendency for bilateralism or zygomorphism (Fig. 22-24).
|
|
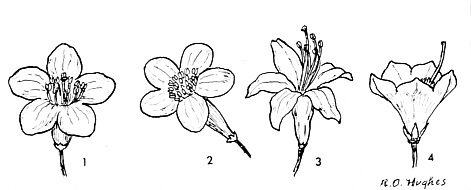
FIG.21. Rhododendron flowers of radial symmetry.
1. R. ferrugineum L. (Ferrugineum ser.) 2. R. polyandrum Hutch. (Maddenii ser.) 3. R. roseum Rehder (Azalea ser.) 4. R. sutchuenense Franch. (Fortunei ser.) |
|
|
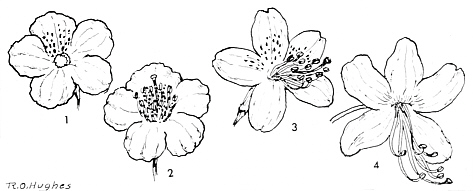
FIG.22. Rhododendron flowers of slightly bilateral symmetry.
1. R. saluenense Franch. (Saluenense ser.), upper corolla lobe with slightly darker markings. stamens and pistil eliminated. 2. Same corolla with stamens and pistils. 3. R. tashiroi Max. (Azalea ser. Tashiroi subser.) upper corolla lobes with markings. 4. R. vaseyi A. Gray (Azalea ser. Canadense subser.). Corolla slightly bilabiate, with orange-red dots at the base of the upper lobes |
|
|
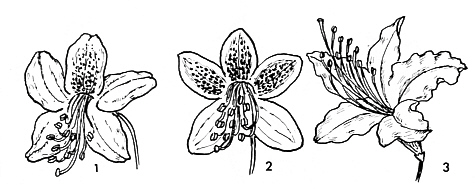
FIG.23. Rhododendron flowers fully bilaterally symmetrical.
1. R. yunnanense Franch. (Triflorum ser.). Bilateral corolla with marked upper petal. 2. R. camtschaticum Pallas (Camtschaticum ser.). Bilateral corolla with marked upper lobes. 3. R. augustinii Hensl (Triflorum ser.). |
|
|
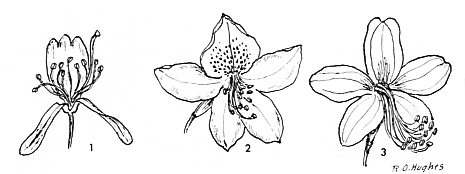
FIG.24. Slightly zygomorphic rhododendron flowers.
1. R. canadense Torrey (Canadense ser.). Bilabiate corolla. The lower tip divided into the narrow-oblong lobes, the upper lip with three short lobes at apex. 2. R. ovatum Maxim. (Ovatum ser.). Corolla slightly zygomorphic, upper lobe with markings. 3. R. stamineum Franch. (Stamineum ser.). Rose or white flowers with yellow blotch, slightly zygomorphic. |
Nectar markings occur most commonly on the upper lobe of the corolla, although less consistent colored spots can be found anywhere on the flower. The upper corolla lobe of bilateral rhododendron flowers is frequently more highly colored than the other petals and it often carries a definite color pattern of clots or blotches of red, purple, brown, yellow, green or even black.
All primitive flowers types of phylogenetically older angiosperms have regular structure and perfect radial symmetry. Such simple floral patterns of successive haplomorphic, actinomorphic, and pleomorphic type classes are the most numerous in present-day floras. Fossil evidence indicates that until the Tertiary period all showy flowers had radially symmetrical shape, without any traces of bilateralism or zygomorphism (LEPPIK 1960, 1963, 1971, 1972, a, b).
Differentiation of rhododendron flowers from the ancestral ericoid type follows the general trend of floral evolution (LEPPIK 1957). The anatomy and morphology of a single rhododendron flower reveal its successive evolution. from an actinomorphic prototype to pleomorphic and stereomorphic levels with tendencies to bilateralism and zygomorphism (Fig. 21-24). In a general sequence of floral evolution the rhododendron's blossoms are on the stereomorphic level (Fig. 21). This evolutionary level is determined by the morphological structure and pollination ecology of the blossoms.
III. Progression of Bilateral Symmetry in the Rhododendron Flowers
Bilateral symmetry is the most remarkable structural progression of floral evolution in general, and in the rhododendron flowers in particular. Beginning with the pleomorphic level. regular flower types became subjected co a profound structural change that directed their further evolution toward bilateralism and zygomorphism. Present knowledge indicates that changes in the shape of the flowers concurred with corresponding progressive changes in the sensory development of their particular pollinators.
In bilateral flowers, we can recognize a remarkable deviation from the regular symmetry of the more primitive actinomorphic flower types and the gradual formation of a zygomorphic pattern as a new style in floral beauty. Ordinarily elementary flower types in lower evolutionary levels have a conspicuously regular shape, in contrast to the bilateral tendencies of more advanced types. Floral bilateralism bridges the wide structural gap between elementary radial symmetry in primitive flowers and the evolved zygomorphism of advanced types (LEPP1K 1972a). Such profound changes in floral structures must be accompanied also by gradual but radical changes and improvements in pollination and fertilization.
Insects may enter a radially symmetrical flower from various directions, and they frequently bypass the stamens and pistils. In bilateral flowers, the insect must approach the flower from a certain direction and must move in a particular way to reach the available source of food. Colored spots on petals indicate the path. In bilateral flowers, stamens and pistils are arranged so that they will touch the visitor's body consecutively and thus make cross-pollination more likely. If protandrous and protogynous flowers are blooming at the same time, crosspollination is the rule.
Depending upon the selective activity of pollinators, bilaterally symmetrical patterns have evolved in a parallel manner in several subgenera and series. Fig. 21 illustrates some radially symmetrical flowers, which can be found in almost every subgenus and series (Fig. 25). Slightly bilateral flowers (Fig. 22) have nectar guides that indicate the shortest route to the nectar deposits in the corolla tube. Fully bilateral flowers (Fig. 23) have markers that lead to nectar deposits.
Truly zygomorphic patterns, such as in labiate, papilionaceous, scrophulariaceous, or orchidaceous flowers, are not known among rhododendrons. Rhododendron flowers have not yet reached the highest specialization and symmetry level in the general trend of floral evolution. On the other hand, several species in various series show slightly zygomorphic floral patterns (Fig. 24). Although members of different series and representatives of various evolutionary lines, they nevertheless developed similar flower types under selective pressure of their common pollinators. Thus, a marked parallelism exists in the floral evolution of various subgenera and series from polymery to pentamery, and from radial symmetry to bilaterlism (Fig. 25).
IV. Phylogenetic and Taxonomic Classification of Rhododendrons
Because of the great number of highly variable species (approximately 850-900) and their wide geographic distribution, the phylogenetic and taxonomic classification of rhododendrons is difficult. Until exact genetic and evolutionary studies on this vast material are completed, any hitherto proposed classification system remains provisional.
For practical purposes species with similar morphological characteristics, though not necessarily related phylogenetically, are joined into definite series. The most complete system of such series of rhododendrons and azaleas was proposed by J. Hutchinson, J. A. Rehder, and H. J. Tagg, and published by STEVENSON (1930, 1947). Comparative anatomical study on the Rhododendroideae was completed by COPELAND (1943) and Cox (1948) and phylogeny mainly by HUTCHINSON (1946) and SLEUMER (1949). Some chemotaxonomic hints on the classification of rhododendrons are made by THIEME and WINKLER (1969). In this paper the system of SLEUMER (1949, 1966) is adapted for the classification of genera, subgenera, and sections. For the arrangement of species under series and subseries (Fig. 25), the listings of STEVENSON (1947), BOWERS (1960), and LEACH (1961) are used.

|
|
FIG. 25. Observable trends of floral evolution in various subgenera (Roman symbols)
and series (Arabic symbols) of rhododendrons. Dots indicate the number of host species for the Chrysomyxa in a series. |
V. Origin and Present Distribution
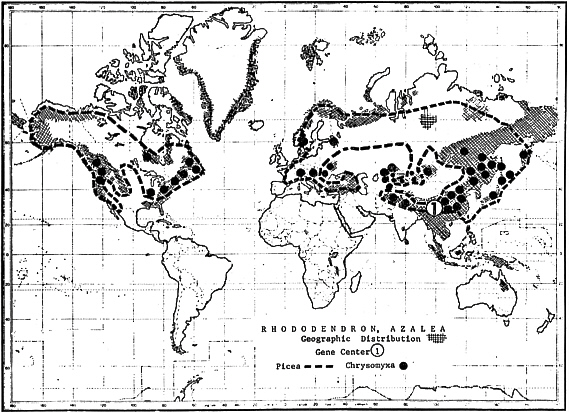
Present distribution of wild rhododendrons is almost entirely restricted to the northern Hemisphere (Fig. 26). The great center of diversity is in southeastern Asia, western China, Tibet, Himalaya, and adjacent areas. Within this small but mountainous territory more than 500 species are found, roughly two-thirds of all known rhododendrons of the world. Some conclusive evidence and fossil records indicate that this area might have been the gene center of the tribe Rhododendreae. From this center ancient members of the group have apparently moved in several directions, adapting themselves to various climatic conditions. Secondary centers of distribution are in southern Asia and tropical islands, including New Guinea, with about 225 species. North Australia has only a single species, probably an immigrant from adjacent islands. Further centers are in Japan with 53 species, northeastern Asia (Korea, Manchuria, Mongolia, Kamchatka) with 14 species. The Arctic with four species and North America with 35 species which are divided into western and eastern regions (Fig.26).
|
|
FIG. 26. Geographic distribution of rhododendrons and azaleas (shaded areas), with suppos-
ed gene center ( 1 ) in east-central Asia. Distribution of the genus Picea is circled with broken line (------) and observed Chrysomyxa infections indicated with dots (·). |
Because of the extermination of original floras in central and north Europe during the Pleistocene glaciation, only four Rhododendron species, ser. Ferrugineum , are known from south European mountains, and five species, ser. Ponticum , from Caucasia. All of these species seem to have migrated to Europe from southeastern Asia after the retreat of ice (LEPPIK 1972b). Common rust flora of European and Asiatic rhododendrons support this view. Such distribution through three northern continents is feasible if the nucleus of the present genus Rhododendron has been formed somewhere on the remnants of the former Laurasian supercontinent. The successive splitting and drifting of North America away from Eurasia could have also carried the corresponding series of rhododendrons to their present places. Several intercontinental bridges over the Pacific Ocean must have favored the movement of plants from eastern Asia to western America and vice versa.
VI. Fossil Remnants of Rhododendrons
Since most rhododendrons are growing in mountains with arid or semiarid climate, their fossilization seldom can occur adequately. Occasionally their hard leaves, wood or fruits are found as imprints or petrified remnants in Tertiary formations.
In the compendium Index of the Paleobotany Library, Smithsonian Institution, Washington, D.C., 40 fossil rhododendron species are listed from Tertiary, and one dubious species from late Cretaceous period. Some entries in this Index, particularly those of the early Tertiary period or Mesozoic era, inevitably need careful re-examination before they could be substantiated. All localities in this Index occur in the central areas of distribution of the present day rhododendrons in Eurasia and North America. No fossils are known from the rhododendrons by newly settled areas, such as New Guinea, Australia, India and Ceylon.
The following list of megafossils of
Rhododendron
species is compiled from the Compendium Index of the Paleohotanical Library, Smithsonian Institution, Washington, D.C., by courtesy of Dr. S. H. Mamay.
| 1. | LATE CRETACEOUS ( 1 species?): | |
| Rhododendron paleoponticum Romanova. | ||
|
|
Kazakhstan, USSR - ref. Romanova, E.V., 1963, Mat. po Isut. Fauny i Flory | |
| Kazakhstana, v. 4, Acad. Nauk, Kazakh. SSR, Inst. zool., p. 105, pl. 2, f. 3. | ||
| 2. | EOCENE (5 species?): | |
| R. megiston Unger, 1950 | Croatia: Radoboj. | |
| R. flosssaturni Unger, | Dalmatia: Monte Promina. | |
| R. celasense Laurent, | France: Dept. de Puy-de-Dóme. | |
| R. flossaturni Unger, | Styria: Parschlug. | |
| R. ponticum L., | France: Crenoble et Chambéry. | |
| 3. | OLIGOCENE (3 spieces): | |
| R. budensis Star, Lower Oligocene, | Hungary: Budapest. | |
| R. chaneyi Brown, | Oregon: Crooked River. | |
| R. celasense Laurent | France: Célas. | |
| 4. | MIOCENE (13 species): | |
| R. alcyonidum Unger, | Hesse, Germany. | |
| R. columbiana Smiley. | Washington. | |
| R. crassum Hollick | Alaska. | |
| R. haueri Hunger (Ettingshausen) | Bohemia: Kutschlin. | |
| R. idahoensis Smith, | Idaho: Thorn Creek. | |
| R. megiston Unger, | Spain: Cerdana. | |
| R. protodilatatum Tanai & Onoe, | Japan: Nigyo-Toge. | |
| R. retusum Goeppert, | Silesia: Schossnitz. | |
| R. rugosum Goeppert, | Silesia: Schossnitz. | |
| R. sagorianum Ettingshausen, | Carniolia: Savine. | |
| R. sierrae Condit, | California: Table Mountain. | |
| R. weaveri (Hollick) Wolfe, | Alaska. | |
| R. uraniae Unger, | Styria: Sotzka. | |
| 5. | PLIOCENE (6 species): | |
| R. columbianum Smiley, Late Pliocene, | Washington. | |
| R. flavum Don., | Poland: Czorsztyna (West Carpathians). | |
| R. gianellana Axelrod, Early Pliocene, | Nevada: Chalk Hills. | |
| R. protodilatatum Tonai & Onoe, | Japan: Nigyo-Toge. | |
| R. protoluteum Kolakovsky, | Georgia, USSR. | |
| R. sierrae Condit, | California: Table Mountain | |
| 6. | PLEISTOCENE (9 species): | |
| R. degronianum Car., | Japan: Honshu. | |
| R . ferrugineum L., | Switzerland: Schneisingen. | |
| R. ponticum L., | Switzerland: Nordalps. | |
| R. ponticum L., | Greece: Skyros. | |
| R. ponticum L., | France: Crenoble et Chambéry. | |
| R. quinquefolium Bisset et Moore | Japan: Kyiishii | |
| R. sebinense Sordelli, | Italy: Varese. | |
| R. tatewakii Tanai | Japan: Shiobara. | |
| R. wadanum Makino, | Japan: Shiobara. | |
| 7. | QUATERNARY (Postglacial, 2 species): | |
| R. lapponicum L. | Great Lakes region. | |
| R. ponticum L. | Greece: Skyros. | |
Less complete are the microfossil records. Entomophilous plants produce relatively small amount of sticky pollen which cannot be carried by wind to lakes or other water basins, where the anemophilous pollen is commonly preserved. The lack of ericaceous pollen in the early Tertiary period can be explained by the predominant entomophily of this family.
VII. Rust Fungi on Rhododendrons
Several species of Chrysomyxa, Pucciniastrum vaccinii (Wint.) Jorst., Puccinia rhododendri Buck., Aecidium rhododendri Barcl., and A. sino-rhododendri M. Wilson, occur on rhododendrons (Table 1). Ten rust species have been reported from Asia (CUUMMINS and LEE LING 1950;KUPREVICZ et TRANZSCHEL 1957), one of which, Chrysomyxa ledi (Alb. & Schw.)d By. var. rhododendri (D. by.) Savile is also found in Europe (GAUMANN 1959) and in arctic North America (SAVILE 1955). In Europe wild rhododendrons and their rusts are scarce (Table 1). Chrysomyxa ledi var. rhododendri and the endemic Puccinia rhododendri are found in the Alps and the Carpathian mountains. North America has two endemic Chrysomyxa species and one Pucciniastrum vaccinii on Vaccinium and Azalea (ARTHUR 1934; SAVILE 1955; (CUMMINS and STEVENSON 1956). All these rusts develop their aecial stage (O, I) on Picea or Tsuga and telial stage (I1, III) on rhododendrons, except Puccinia rhododendri (I1, III) and several autoecious forms with incomplete life cycle.
| Table I. Distribution of Rust Genera on Rhododendrons | ||||
| RUSTS AND HOSTS | ASIA | EUROPE | AMER. | ARCTIC |
| 1. Chrysomyxa ledi var. rhododendri O, I on Picea; | ||||
| II, III on Rhododendron | X | X | X | |
| 2. Ch. dieteli III on Rhododendron | X | |||
| 3. Ch. expansa III on Rhododendron | X | |||
| 4. Ch. hirnalensis III on Rhododendron | X | |||
|
5. Ch. kornarovi III on Rhododendron |
X | |||
| 6. Ch. piperiana O, I, II, III on Rhododendron | X | |||
| 7. Ch. roanensis O, I, II, III on Rhododendron | X | |||
| 8. Ch. succinea O, I, II, III on Rhododendron | X | |||
| 9. Ch. taghishae III on Rhododendron | X | |||
| 10. Ch. tsugae-yunnanensis III on Tsuga | X | |||
|
11.
Pucciniastrum vaccinii
O, I, on
Tsuga;
II, III on Rhododendron |
X | |||
| 12. Aecidium rhododendri I on Rhododendron | X | |||
| 13. A. sino-rhododendri I on Rhododendron | X | |||
| 14. Puccinia rhododendri III on Rhododendron | X | |||
| TOTAL | 10 | 2 | 3 | 1 |
Such unequal dispersal pattern indicates that several groups of conifer rusts, in different geological epochs, evaded rhododendrons on Eurasian and North American continents. The largest rust variety,
Chrysomyxa ledi
var.
rhododendri
, occurs on phylogenetically older rhododendron series in the mountains of central Asia. This area is assumed to be the gene center of rhododendrons and the birth place of some rhododendron rusts (Fig, 26). Since the heteroecious rhododendron rusts are derived from the older
Ch. ledi
on
Ledum
and
Picea
, all three host genera must have grown in close neighborhoods. From this original area rhododendron rusts have dispersed with their hosts to all directions, including the Arctic and North America.
After the separation of North America from Eurasia and during the Pleistocene glaciations, the original area of rhododendrons became disrupted. American rhododendrons with their rusts could move gradually southward or survive in ice-free pockets. They could return to the north after withdrawal of the glaciers. During these presumable dislocations of hosts, differentiation of endemic
Ch. roanensis
and
Ch. piperiana
very likely took place from an ancestral rust stock on
Ledum
and
Picea
. SAVILE (1950:323) for instance, suggests a close relationship between
Ch. ledicola, Ch. empetri, Ch. roaneusis,
and
Ch. piperiana
. The common Eurasian rust,
Ch. ledi
var.
rhododendri
, appears to be distributed only on subarctic
Rh, lappopicum
, in Canada, nearly from coast to coast (SAVILF 1955). This host with its rust reached its circumpolar distribution obviously in post-glacial time.
Many Eurasian rusts on cultivated rhododendrons in America are obviously imported with nursery stocks. These rusts commonly overwinter as dormant mycelium in rhododendron leaves, thus bypassing plant quarantine inspection at the port-of-entry stations. For detection and interception of these foreign rusts a post-entry surveillance is unavoidable.
Adaptation of
Pucciniastrum. vaccinii
(Wint.) Jorst. to azaleas occurred probably in America only. Although this rust is distributed on
Vaccinium
throughout the northern hemisphere, its occurrence on rhododendrons has not been reported from Eurasia.
In Europe, however, the fate of rhododendrons and their rusts during the Pleistocene glaciation was different from that in North America. The path of south-moving plants was abruptly cut off by other glaciers moving from opposite directions from south and central European mountains, such as Pyrenees, Alps, and Carpathian ranges. This might be one reason why rhododendrons and their rusts are scarce in Europe. There are only two rust species: Eurasiatic
Chrysomyxa ledi
var.
rhododendri
and endemic but rare
Puccinia rhododendri
. The last species belong to the group of modern rusts with uncovered telia and short, fragile pedicals. It might be a relatively recent, post-glacial adaption among rhododendron rusts.
| List of Rusts Species Recorded on Rhododendrons | |
| 1. |
Chrysomyxa ledi By., var. rhododendri (D By.) Savile. O, I on Picea; II, III on Rhododendron, Azalea |
| Rhododendron aberconwayi Cowan (ser. Irroratum, Malay, cult in USA). | |
| R. arboreum Sm. subsp. cinnamomeum Wall. (ser. Arboreum, Himalaya, cult. in USA) | |
| R. augustinii Hems]. (ser. Triflorum, Szechuan, cult. in USA). | |
| R. brachyanthum Franch. (ser. Glaucophyllum, Yunnan, cult. in USA). | |
| R. bureavii Franch. (ser Taliense, Yunnan, cult. in USA). | |
| R. burmanicum Hutch. (ser. Maddenii, Burma, cult. in USA). | |
| R. calostrotum Balf. f. & Ward (=R. riparium Ward) (ser. Salunenense, Burma, cult. in USA). | |
| R. camtschaticum Pallas (ser. Camtschaticum, Alaska, Okhotsk, Japan, cult. in USA). | |
| R. campylogynum Franch. (ser. Campylogynum, Yunnan, cult. in USA). | |
| R. carolinianum Rehd. (ser. Carolinianum, southern Europe, cult. in USA). | |
| R. catawbiense Michx. (ser. Ponticum, cult. in USA). | |
| R. charitopes Balf. f. & Farrer (ser. Glaucophyllum, Burma, cult. in USA). | |
| R. chrysanthum Pallas (ser. Ponticum, Siberia, Manchuria, Japan, cult. in USA). | |
| R. dauricum L. (ser. Dauricum, North Asia, Japan). | |
| R. decorum Franch, (ser. Fortunei, Yunnan, Szechuan). | |
| R. degronianum Carriere (=R. pentamerum Maxim., ser. Ponticum, Japan). | |
| R. fauriei Franch. (ser. Ponticum, Japan, cult. in USA). | |
| R. faberi Hemsl. (ser. Taliense, Szechuan). | |
| R. ferrugineum L. (ser. Ferrugineum, southern Europe). | |
| R. fimbriatum Hutch. (ser. Lapponicum, Yunnan, cult. in USA). | |
| R. forrestii Balf. f. MS. (=R. repens cult. var. 'Ethel'), (ser. Neriiflorum, Yunnan, Tibet, cult. in USA). | |
| R. glomerulatum Hutch. (ser. Lapponicum, Yunnan, cult. in USA). | |
| R. griersonianum Balf. f. & Forrest (ser. Auriculatum, Yunnan, cult. in USA). | |
| R. hemitrichotum Balf. f. & Forrest (ser. Scabrifolium, Szechuan, cult. in USA). | |
| R. hippophaeoides Balf. f. & W. W. Sm. (ser. Lapponicum, Yunnan, cult. in USA). | |
| R. hirsutum L. (ser. Ferrugineum, Europe, cult. in USA). | |
| R. hormophorum Balf. f. & Forrest (ser. Triflorum, Szechuan, cult. in USA). | |
| R. impeditum Balf. f. & W. W. Sm. (ser. Lapponicum, Yunnan, cult. in USA). | |
| R. imperator Hutch. & Ward (ser. Uniflorum, Burma, cult. in USA). | |
| R. indicum Sweet (=R. lateritium Planch.), (ser. Azalea, subser. Obtusum, Japan). | |
| R. kaempferi Planch. (=obtusum Planch, var. kaempferi Wils.), (ser. Azalea, sub ser Obtusum, | |
| West Siberia, Far East, Japan) | |
| R. kiusianum Makino (ser. Azalea, subser. Obtusum, Japan). | |
| R. kotschyi Simk. (=R. myrtifolium Schott & Kotschy, not Lodd), (ser. Ferrugineum, Hungary). | |
| R. lapponicum L. (ser. Lapponicum, Canada). | |
| R. ledebourii Pojark. (ser. Dauricum, Altai, Sayan, collected also from Tulun (Irkutskaya oblast) | |
| and Barnoul (Altai area) by J. L. Creech, July 1971). | |
| R. linearifolium Sieb. & Zucc. (ser. Azalea, subser. Obtusum, Japan, Canada). | |
| R. maximum L. (ser. Ponticum, cult. in Norway). | |
| R. micranthum Turez. (ser. Micranthum, China). | |
| R. molle G. Don. (ser. Azalea, subser. Luteum, China, cult. in USA). | |
| R. mucronatum G. Don. (ser. Azalea, subser. Obtusum, China). | |
| R. occidentale A. Gray (ser. Azalea, subser. Luteum, North America, Florida). | |
| R. odoriferum Hutch. (ser. Maddenii, Tibet, cult. in U.S.A.). | |
| R. oldhamii Maxim. (ser. Azalea, subser. Obtusum, Formosa). | |
| R. parvifolium Adams (ser. Lapponicum, Japan). | |
| R. pemakoense Ward (ser. Uniflorum, Tibet, cult. in Canada and USA). | |
| R. pseudochrysanthum Hayata (ser. Barbatum Taiwan, cult. in USA). | |
| R. racemosum Franch. (ser. Virgatum, Yunnan, cult. in USA). | |
| R. radicans Balf. f. & Forrest (ser. Saluenense, Tibet, cult. in USA). | |
| R. saluenense Franch. (ser. Saluenense, Yunnan, cult. in USA). | |
| R. scabrifolium Franch. (ser. Scabrifolium, Yunnan, cult. in USA). | |
| R. sphaeroblastum Balf. f. & Forrest (ser. Taliense, Szechuan, cult. in USA). | |
| R. tsangpoense Hutch. (ser. Glaucophyllum, Tibet, cult. in England). | |
| R. uniflorum Hutch. & Ward (ser. Uniflorum, Tibet, cult. in USA). | |
| R. zaleucum Balf. f. & W. W. Sm. (ser. Triflorum, Yunnan, cult. in USA). | |
| 2. | Chrysomyxa dieteli Syd. III |
| Rhododendron arboreum Sm. (ser. Arboreum, Himalaya, China). | |
| R. oldhamii Maxim. (ser. Azalea, subser. Obtusum, Taiwan). | |
| R. lepidotum Wall. (ser. Lepidotum, Himalaya, Kashmir, Tibet). | |
| R. subsessile Rendle (ser. Azalea, subser. Obtusum, Philippines). | |
| R. trichocladum Franch. (ser. Trichocladum, China). | |
| 3. | Chrysomyxa expansa Diet. III |
| Rhododendron decorum Franch. (ser. Fortunei, Yunnan). | |
| R. erythrocalyx Balf. F. & Forrest (ser. Thomsonii, Yunnan). | |
| R. fauriei Franch. (ser. Ponticum, Japan). | |
| R. metternichii Sieb. & Zucc. (ser. Ponticum, Japan). | |
| R. morii Hayata (ser. Barbatrum, Taiwan). | |
| R. selense Franch, (ser. Thomsonii, Szechuan). | |
| R. wardii W. W. Sm. (ser. Thomsonii, Yunnan, Szechuan). | |
| 4. | Chrysomyxa himalensis Barcl. III |
| Rhododendron arboreum Sm. (ser. Arboreum, Himalaya, cult. in India). | |
| R. campanulatum D. Don. (ser. Campanulatum, Himalaya, cult. in India). | |
| R. hodgsonii Hook. F. (ser. Falconeri, Himalaya). | |
| R. vellereum Hutch. MS., (ser. Taliense, Tibet). | |
| 5. | Chrysomyxa komarovii Tranz. III |
| Rhododendron dauricum L. (ser. Dauricum, West Siberia) III. | |
| 6. | Chrysomyxa piperiana (Arth.) Sace. & Trott, ex Cumm. |
| Rhododendron californicum Hook. (ser. Ponticum, III, in coastal region of northwestern United States). | |
| 7. | Chrysomyxa roanensis (Arth.) Arth. O, I on Picea rubens; II, III on Rhododendron |
| Rhododendron carolinianum Rehd. (=Rh. punctatum Ker, ser. Carolinianum, USA, Tennessee). | |
| R. catawbiense Michx. (ser. Ponticum, USA, Tennessee). | |
| 8. | Chrysomyxa succinea (Sace.) Tranz. (=Clr. Alphina Hirats.) O, I, II, III Picea jezoensis |
| (Sieb, & Zucc.) Carr. (=P. ajanensis Fisch. ex Carr.) O, I. | |
| Rhododendron chrysanthum Pallas (=R. aureum Georgi), (Ser. Ponticum, Siberia and Far East) II, III. | |
| R. nankotaisanense Hayata (ser. Barbatum, Taiwan) II, III. | |
| 9. | Chrysomyxa taghishae Balfour-Browne (1955) III |
| Rhododendron sp. (dwarf), on leaves, Nepal. | |
| 10. | Chrysomyxa tsugae-yunnanensis Teng. III |
| Tsuga yunnanensis (Franch.) Bark. (China) III. | |
| 11, | Puccinia rhadodendri Fuck. III |
| Rhododendron ferrugineum L. R. hirsutum L. and R. kotschyi Simk. (ser. Ferrugineum, in eastern | |
| Alps and Carpathian mountains). | |
| 12. | Pucciniastum vaccinii (Wint.) Jrst. (= P. myrtilli Arth.) O, I on Tsuga; II, III on Azalea |
| Rhododendron canescens Sweet (=Azalea c. Michx.) (ser. Azalea, subser, Luteum, | |
| in east coast of North America) | |
| R. calendulaceum Torrey (=Azalea nudiflora L.) (ser. Azalea, subser. Luteum, | |
| east coast of North America) | |
| R. viscosum Torrey (=Azalea v. L.) (ser. Azalea subser. Luteum, in east coast of North America). | |
| Imperfect Rusts on Rhododendrons | |
| 13. | Aecidium rhododendri Barclay I |
| Rhododendron campanulatum D. Don. (ser. Campanulatum, Himalaya). | |
| 14. | Aecidium sino-rhododendri M. Wilson I |
| Rhododendron calvescens Balf. f. & Forrest (ser. Thomsonii, Tibet). | |
| R. dryophyllum Balf. f. & Forrest (ser. Lacteum, Yunnan). | |
| R. schizopeplum Balf. f. & Forrest (ser. Taliense, Szechuan). | |
| R. sphaeroblastum Balf. F. & Forrest (ser. Taliense, Szechuan). |
VIII. Continental Drift
|
|
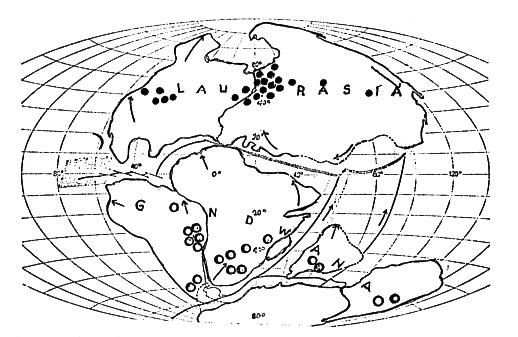
FIG.27. The northern group of continents has split away from southern
Gondwanaland at the end of Triassic period, about 180 million years ago (after 20 million years of drift). In Jurassic, North American separated from Laurasia and Gondwanaland broke up into South America, India and Antarctica-Australia. (After DIETZ and HOLDEN, 1970; courtesy of the Scientific American. ) Fossil finds of northern conifers northern Pinaceae, Taxodiaceae and Cupressaceae are contrasted with southern Podocarnaceae and Araucariaceae (o) according to FLORIN (1963). |
Recent geological evidence indicates that the original land mass Pangaea split during the Permian period into two supercontinents: Laurasia in the north and Gondwanaland in the south (Fig. 27). Both supercontinents were separated by the Tethys Sea, the present Mediterranean being its remnant. Laurasia was composed of present Eurasia and North America; Gondwanaland of present Africa, India, Australia, Antartica and South America.
The drifting of supercontinents and formation of the Tethys Sea became an important phytogeographic factor, separating the northern and southern floras. Occasionally water currents carried seeds over the Tethys Sea, favoring exchange of plants between southern and northern floras (EDWARDS 1955).
Although the time and place of origin of angiosperms remains tentative, it is commonly agreed that the most angiospermous families were differentiated during the Cretaceous period and most genera in the Tertiary period. Ancestors of the present Ericaceae were accordingly divided into northern and southern genera. The genus
Rhododendron
(including
Azalea)
was probably formed on remains of the Laurasian supercontinent during the Tertiary period when both supercontinents were separated from each other by the Tethys Sea. All its members are distributed on northern continents only (Fig. 26). There are no wild rhododendrons in Africa. Central and South America, or Polynesia. Only a single species,
R. lochiae
F. Muell., grows in northern Australian mountains. This species is related to the Javanese rhododendrons and is obviously a more recent escape from adjacent islands (HUTCHINSON 1947:88). The most striking fact is the rarity of wild rhododendrons on the Indian peninsula, which is considered to be part of former Gondwanaland. On the other hand, the slopes of the Himalayan mountains in northern India and adjacent Kashmir, Nepal, Sikkim, and Bhutan are rich in wild rhododendrons. These areas belong to the Asian mainland of the Laurasian supercontinent and were separated from the Gondwanaland and India proper by the immense Tethys Sea. Thus, the geographical distribution of present rhododendrons agrees with the theory of continental drift.
One Himalayan species,
R. arboreum
Sm., has recently migrated southward, forming a subsp.
nilagiricum
Zenker, in the mountains of Madras, in southeast India, and another subsp.
zeylanicum
Hort. ex Loun. in the mountains of Ceylon (HUTCHINSON 1947:90).
R. indicum
Sweet. (
Azalea indicum
L.) is an accidentally misnamed Japanese species, as are the numerous "Indian Azaleas" of florists which actually are of Chinese origin (BOWERS 1960:259).
Additional evidence for the drift theory is provided by heteroecious rust fungi that complete their life cycle on two alternate hosts (Fig. 26). Several species of
Chrysomyxa
and a
Pucciniastrum
form their telial stage on rhododendrons, but their actual stage on
Picea
or
Tsuga.
Ancestral rhododendrons, must have lived, therefore, in an area where spruces or hemlocks were common, possibly in early spruce forests.
Picea, Tsuga, Chrysomyxa,
and
Pacciniastrum
had a northern origin (LEPPIK 1972 b, 1973).
Rust infection certainly does not prevent rhododendrons from moving out of conifer forests and settling outside of spruce or hemlock areas (Fig. 26). Presently, rhododendrons prefer deciduous or mixed forests to conifer forests and are frequently associated with
Myrtillus
and
Kalmia.
If separated from its conifer host, rust fungi commonly adjust to autoecism and restrict their life cycle to telial stage only. Such is the case of
Chrysomyxa dieteli, Ch. himialensis, Ch. expansa,
and
Puccinia rhododendri.
Both
Picea and Tsuga
are phylogenetically older than the genus
Rhododendron.
The fossils of
Picea
go back to late Cretaceous, about 100 million years ago (FLORIN 1963). The first rhododendron fossils are known from lower Tertiary (late Paleocene) about 50 million years ago. The origin of
Chrysomyxa
can be connected with the geological period when
Picea
and
Tsuga
were separated from the Pinoideae, approximately 100 million years ago. Closely related
Picea
and
Tsuga
serve as the primary hosts and as the center of biogenic radiation for the genus
Chrysomyxa.
From this center the spruce rust has radiated to rhododendrons and various other genera of Ericaceae, Pyrolaceae, Empetraceae, and Aquifoliaceae of the orders Ericales and Sapindales (LEPPIK 1965, p. 13). All these plants grow in spruce forests, indicating that at one time rhododendrons also must have been associated more closely with spruces than they are today.
IX. Pollination and Pollinators
So-called "bumblebee flowers" of rhododendrons are protandrous and brightly colored, with nectar secreted by an annular ring at the base of the ovary. Stamens are hypogynous and filaments are provided with erect hairs for the protection of nectar against rain. Tetrahedrons pollen grains are squeezed out from pollen sacs through the apical pores, either by the anther shriveling or by activity of pollinators (FAECRI and van der PIJL 1966; p. 17: KUGLER 1970, p. 237). All the pollen in one sac comes out in a mass that is held together by viscid strands (BOWERS 1930, p. 132; 1960, p. 138).
In order to get at the nectar, insects (bumble bees and butterflies) must creep over the stamens and stigma into the base of the blossom. If the flower is in its first (male) stage, the insect body is dusted with viscous pollen, which will be transferred to the stigmas of flowers in the second (female) stage. In some species the longest stamens project beyond the stigma, causing automatic self-pollination if insect visits fail.
Legitimate pollinators of rhododendrons and azaleas are considered to be wild bees, honey bees, bumble bees, and butterflies. In addition, many occasional visitors, such as beetles, flies, wasps, ants, and others are also observed on rhododendron flowers. Bird pollination of rhododendrons is reported from Malesia (SLEUMER 1966: 476).
Rhododendron retusum
(B1.) Berm. has been seen visited by honey birds,
Aethopyga eximea,
and
Melidectes fuscus
(De Vis).
In spite of the rather simple pollen presentation mechanism of the rhododendron flowers, the process of pollination itself is fairly complicated, and varies from species to species. In more elementary tubular flower types, nectar is hidden in the bottom or corolla tube and is accessible only to visitors with proboscises long enough to reach it. More advanced rhododendron flowers have a tendency to become bilateral and zygormorphic with colored markings commonly on one or three upper corolla lobes (Figs. 22-23). The upper lobe is frequently more intensely colored than the other parts of the corolla and that lobe often exhibits a definite color pattern of clots or blotches (Fig. 24). Dots are yellow, green, red, purple, brown, or black, contrasting to the colors of the corolla.
Dots and blotches on corolla lobes serve as nectar guides to pollinators. indicating how to reach the nectar deposits. Bees, bumble bees, and other hymenopterous pollinators learn to recognize these signs at a distance and approach the upper half of the horizontal flower. They follow the way indicated by colored clots or blotches until they reach the nectar. Insect weight causes the stamens and style to curve upward, contacting the ventral side of the insect body. Other species develop fairly long stamens and pistils that are curved upward so that they can touch the back of pollinators. In the first (male) stage the style is shorter than the stamens and the stigma is immature. The anthers dehisce apically by means of two openings and dust the ventral surface of visitors by viscous pollen. In the second (female) stage the style elongates and the stigma attains its full length above the wilted and withered stamens. In this stage the insects touch the style, covering it with pollen brought from other flowers.
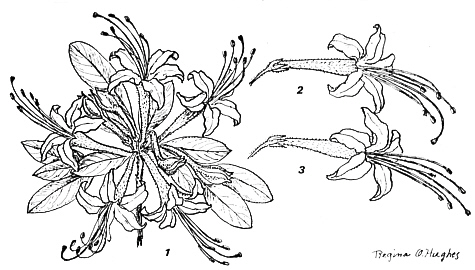
Pollination of R. nudiflorum (L.) Torr.
|
|
FIG.28.
Rhododendron nudiflorum
(L.) Torr. (=Azalea n. L.).
- 1. A flowering umbel of 6-12 flowers (reduced). - 2. First stage of anthesis - pistil is curved downward. - 3. Second stage of anthesis - pistil is joined with anthers (almost natural size). |
Pinxter-flower,
Rhododendron nudiflorum
(L.) Torr. (Azalea n. L.), found in the forests of eastern North America, is one of the earliest species to bloom (April-May). It is characterized by a radial or somewhat bilateral 5 lobed (2+3) corolla about 4 cm across, with a narrow funnel-form tube 2-2.5 cm long (Fig, 28). Nectar is secreted by an annual ring at the base of the ovary. It is accumulated in the lower part of the tube and therefore is accessible to insects with sufficiently long proboscises. Slightly fragrant pinkish or whitish flowers, 4-5 cm in diameter, are borne in 6-12 flowered umbels. The flowering shrubs (1-2 m high) are tightly covered with whitish clusters, which make the plant visible from a distance. The shrubs prefer acid soils and are associated with
Vaccinium
spp. and, less frequently, with
Kalmia
in mixed forests.
A flower bears 5 stamens about equal length (5-6 cm), assembled in a slightly upward curved loose bundle. The pistil is 6 cm long, first curved downward, later stretched upward (Fig. 26). In the first stage of anthesis, the downward-curved pistil separates the stigma from the anthers, thus preventing self pollination. Alighting insects rub off the pollen tetrads united by threads of viscin from the anthers. In the second stage of anthesis, the sticky stigma is brought into line with the anthers, ready to catch alien pollen from visitors' bodies. Ultimately, if crosspollination fails, self-pollination may take place.
Insect visitors to
R. nudiflorum
were recorded in the Greenbelt forest, Greenbelt, Maryland, in May 1972. In spite of its having a typical bumble bee flower, only a few bumble bees were observed. Pollen-collecting wild bees,
Doros aequalis
(Loew) Diptera, Syrphidae, (det. by L. V. Knutson), were the predominant visitors and successful pollinators throughout the blooming season. They made up 85 percent of the total visitors recorded in this locality. Upon alighting on the flowers, these bees did not try to reach the nectar, but climbed up the bundle of stamens to anthers, shaking pollen out of the thecae and storing it in their pollen baskets. In the first stage of anthesis, bees could not touch the stigma, which was curved downward. In the second stage, however, the stigma was close to the anthers and could easily catch the alien pollen from the bees' bodies.
Brightly colored butterflies were second in number of total visitors (about 10 percent). They approached the flower from the side, alighted on the corolla, and sucked nectar with their long proboscises. If the flower was in its first stage of anthesis, the butterflies became covered with pollen, which was carried to the next flower. If the flower was in the second stage of anthesis, sticky stigma contacted alien pollen on the bodies of the butterflies. As a rule, the butterflies moved from flower to flower and from plant to plant and thus caused cross-pollination.
Bumble bee queens (5 percent of all pollinators) approached the flowers mainly from the upper part of corolla, sucking nectar from the flower tube. In the first stage of anthesis their large bodies touched only the anthers; in the second stage they touched the stigma.
In addition to these legitimate pollinators, many illegitimate visitors, including honey bees, wasps, flies (for instance,
Condylostylus caudatus
(Wiedemann), (det. by G. C. Steyskal) ), and ants, were observed. These visitors could neither reach the nectar nor pollen nor cause pollination. Honey bees, for instance, were frequently seen walking outside of the corolla tube, sucking a few droplets of extra floral sugar, or looking for holes in order to steal the nectar. Holes are commonly pierced in flower tubes by short-tongued bumble bees that cannot reach nectar from above the corolla. Such holes were not found in
R. nudiflorum.
However, it is feasible that pollen-collecting honey bees can cause pollination, as do wild bees.
Summary
Evolution of rhododendrons and azaleas is studied in relation to their geographic distribution, fossil records, and observable trends of floral evolution, and in correlation with the dispersal of their heteroecious rusts. According to this study, the center of distribution of the tribe Rhododendreae (cox 1948) lies in the south-central Asia, where the greatest number of phylogenetically older species still exists (Fig. 26). From this center they have spread to all parts of the former Laurasian supercontinent, including present Eurasia and North America. Their southward drive evidently was cut off by the intercontinental Tethys Sea, excluding them from the Gondwanaland. As a result, there are no wild rhododendrons in Africa, Central and South America, and Polynesia. Australia and India moved during the continental drift closer to Asian mainland, and one rhododendron species spread to each of these areas, very likely from Malaya and Himalaya.
Floral structure of rhododendrons suggests that the hymenopterous visitors could have been the principal pollinators of these plants during the evolution of their flowers types. The main evolutionary trend of rhododendron flowers proceed, from the gamopetalous ericoid type toward bilateralism and zygomorphism and from polymery toward pentamery. In some special cases, such as
R
.
carringtoniae
F. Muell of New Guinea (Fig. 22:2), only long-tongued butterflies are able to reach nectar. Bird pollination of rhododendrons is reported from Malesia (SLEUMER 1966:476).
There are many species with generalized, broad, funnel form flowers, accessible to many visitors but pollinated mainly by hymenopterous insects. There are other species with long, narrow flower tubes, accessible only to long-tongued bumble bees and butterflies.
Heteroecious rhododendron rusts of the genus
Clirysomyxa
need
Picea
and
Tsuga
as alternate hosts. They indicate that rhododendrons once must have grown in close association with spruces and hemlocks. These rusts and their alternate hosts are restricted exclusively to the northern hemisphere and have evolved necessarily in unison.
Acknowledgments
| The author wishes to express his appreciation to A. E. Kehr. L. T. Hickey, H. F. Winters, E. E. Terrell, |
| C. R. Gunn, and F. A. Uecker for many comments and suggestions. Regina O. Hughes prepared the line drawings. |
| References |
| ARTHUR. J. C. 1934. Manual of the rusts in United States and Canada. Lafayette. Ind. 438 pp. Supplement |
| by G. B. Cummins, New York. 1962, 24 pp. |
| BOWERS, C. G. 1936. Rhododendrons and azaleas, their origin, cultivation and development. The Macmillan |
| Co.. New York, 549 pp. 2d edition 1960, 525 pp. |
| COPELAND, H. F. 1943. A study. anatomical and taxonomic, of the genera of Rhododendroideae. |
| Amer. Midl Nat. 30: 5.33023. |
| COX H. T. 1948. Studies in the comparative anatomy of the Ericales. I. Ericaceae- subfamily Rhododendroideae. |
| Amer. Midl. Nat. 39: 220-245. |
| DIETZ, R. S. and HOLDEN, T. C. 1970. The breakup of Pangaea. Sci. Amer. 223: 30-41. |
| CUMMINS. G. B., and LEE LING. 1950. An index of the plant rusts recorded for continental China and |
| Manchuria. Plant Dis. Rep. Suppl. 196: 520-556. |
| CUMMINS, G. B., and STEVENSON, J. A. 1956. A check list of North American rust fungi (Uredinales). |
| Plant Dis. Rep. Supplm. 240, 193 pp. |
| EDWARDS, W. N. 1955. The geographical distribution of pas: floras. Advanc. of Science 12: 165-176. |
| FAEGRI, K. and L. van der PIJL. 1966. The principles of pollination ecology. Pergamon Press. 248 pp. |
| 2nd edition 1970. |
| FLORIN, R. 1963. The distribution of conifer and taxad genera in time and space. Acta Hort. Bergiani |
| 20 (4): 121-312. Uppsala. |
| GAUMANN, E. 1959. Die Rostpilze Mitteleuropas mit besonderer Berucksichtigung der Schweiz. Buehler, |
| Berw. 1407 pp. |
| HICKY, L. T. 1972. Smithsonian Institution. Washington. D.C. (personal letter) |
| HUTCHINSON, J. 1946. Evolution and classification of rhododendrons. The Rhododendron Year Book 1946 |
| 42-47 London. |
| - 1947. The distribution of rhododendrons. The Rhododendron Year Book 1947: 87-98. London. |
| KEHR. A. E. 1972. Research - What's new in '72. Quart. Bull. Amer. Rhodod. Soc. 26: 223-234. |
| KUGLER, H. 1970. Blutenokologie 2. Auflage. G. Fischer Verlag. Stuttgart, 345 pp. |
| KUPREVICZ, V. Th., et TRANZSCHEL, V. G., 1957. Flora plantarum cryptogamarum URSS, vol. IV, |
| Fungi (1). Uredinales, fasc. I, familia Melampsoraceae. Acad. Scient. URSS, Mosqua, Leningrad, 419 pp. |
| LEACH, D.C. 1961. Rhododendrons of the world and how to grow them. C. Scribner's Sons, New York, 544 pp |
| LEPPIK, E. E. 1957. Evolutionary relationship between entomophilous plants and anthophilous insects. Evolution, |
| 11: 466-481. |
| - 1960. Early evolution of flower types. Lloydia 23: 72-92. |
| - 1961. Evolution of roses vs. breeding. Amer. Rose Annual 46: 59-60, 102-106. |
| - 1963. Fossil evidence for floral evolution. Lloydia 26: 91-115. |
| - 1965. Some viewpoints on the phylogeny of rust fungi. V. Evolution of biological specialization. |
| Mycologia 57: 6-22. |
| - 1971. Paleontological evidence on the morphogenic development of flower types. Phytomorphology |
| 21: 164-174. Delhi, India. |
| - 1972a. Origin and evolution of bilateral symmetry in flowers. Evol. Biol. 5: 49-85. |
| - 1972b. Post-glacial migration of rust fungi to north Europe. Ann. Bot. Fennici 9: 65-90. |
| - 1973. Origin and evolution of conifer rusts in the light of continental drift. Mycopath. et Mycol. Applic. (in print). |
| NITZELIUS, T. G. 1970. Rhododendron brachycarpum D. Don ex G. Don ssp. tigerstedtii eine neue Unter |
| - art. Deutsche Baumschule 22 (7): 207-212. |
| - 1972. Rhododendron brachycarpum D. Don ex G. Don ssp. tigerstedtii , a new subspecies. Quart. |
| Bull. Amer. Rhodo. Soc. 26 (3): 165-168. |
| SAVILE, D. B. O. 1950. North American species of Chrysomvxa , Canad. Jour. Res. C. 28: 318-330. |
| - 1955. Chrysomrxa in North America, additions and corrections. Canad. J. But. 33: 487-496. |
| SLEUMER, H. 1949. Ein System der Gattung Rhododendron L. (A systematic treatment of the genus |
| Rhododendron) In Engler's Botany Jahrb. 74:511-553. |
| - 1966. Ericaceae. In C. G. G. J. van Steenis. Flora Malensiana Ser. I, vol. 6, part 4: 469-668. Leyden, Holland. |
| STEVENSON, J. B. 1930, 1947. The species of Rhododendron The Rhododendron Society, London. 861 |
| pp. 2d eition 1947, 861 pp. |
| THIEME, H. and WINKLER, H. J. 1969. Chemotaxonomic significance of rhododendron. Occurrence in |
| "Ericaceae" Rhododendron species (German). Pharmazie 24 (11): 703. |
| WILSON, M. 1921. Some fungi from Tibet. Notes Roy. Bot. Gard. Edinburgh 12: 261-263. |