Rhododendron lapponicum in Wisconsin
Distribution, Ecology, and Propagation Attempts
By Robert H. Read
Bureau of Research, Wisconsin Department of Natural Resources, Madison, Wisconsin
and Conrad J. Wrzesinski Middleton, Wisconsin
Introduction
The 1.3x 10
5
km
2
(50,000 square mile) "Driftless Area" of southwestern Wisconsin, northeastern Iowa, northwestern Illinois and southeastern Minnesota has long been of special botanical interest due to the presence of a number of vascular plants widely disjunct from their main range, or which are regional endemics (having the major portion of their range in this area). Most of these interesting species are found in special habitats, mainly cliffs, a fact which has caused considerable discussion among plant geographers as to whether the plants occur in this (Wisconsin age) un-glaciated region as relics of past floras, or rather because habitats have been available over thousands of years for which long distance reception of propagules may have been possible.
Whatever the history for the establishment of these species in the Driftless Area, their present occurrence here, often far removed from the closest localities of the same or allied species, reflects very special conditions that have persisted over a very long time. One of these species, Lapland rosebay (
R. lapponicum
; Ericaceae) is the subject of this paper, in which the distribution, ecology and initial attempts on propagation of plants occurring in the Driftless Area will be discussed.

|

|
|
Photo 1.
R. lapponicum
in flower at the Dells of the
Wisconsin River, Columbia County, Wisconsin on 28 April 1977. The peak flowering period of the Wisconsin Cliff-dwelling plants is usually several weeks earlier than that of plants in alpine and arctic habitats to the far north and east. |
Photo 2. The population of
R. lapponicum
on a sheer
north-facing sand-stone cliff along the Kickapoo River (Vernon County) in southwestern Wisconsin is extremely small, consisting of no more than nine clumps (probably each an individual plant). This location was discovered in September, 1976, and the plants first photographed here in flower on 28 April 1977. |
|
|
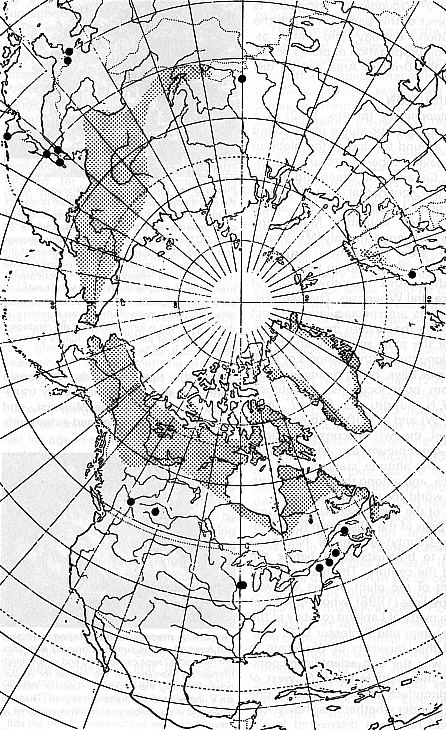
Figure 1: Northern polar projection showing the distribution of
Lapland rosebay ( R. lapponicum ). Shaded portion shows the main range of the species. Note the disjunct locations (indicated by the dots) in eastern North America in the Gaspé, New England and Wisconsin. Map adapted from Hultén (1958) |

|
|
Photo 3. At the Dells of the Wisconsin River, the
entire population of R. lapponicum is restricted to the top edge of this west-facing sandstone cliff, shown here in the full afternoon sun (3:30 p.m. CDT) on 3 May 1977. A constant seepage at the cliff summit and the direct position of the plants about 4 m above the Wisconsin River helps keep the relative humidity consistently high in the vicinity of the plants on this otherwise xeric appearing cliff. A resort has been in existence for many years with- in several meters of the summit, but is fenced off from the rhododendron because of the dangerous nature of the indented, flaky, sandstone cliffs. |

|
|
Photo 4. A precipitous, north-facing sandstone cliff
forested on the summit primarily with eastern hem- lock ( Tsuga canadensis ) and yellow birch ( Betual alleghaniensis ) is the habitat for R. lapponicum along the Kickapoo River in Wisconsin's un-glaciated southwestern region. The very small and inaccessible population is scattered in crevices over the approximately 15 m tall cliff. |
Distribution
R. lapponicum
is a wide-ranging arctic-alpine plant, found mainly around the top of the globe in Scandinavia, Siberia, Japan, Korea, Alaska, Canada, and Greenland. In the continental United States, it has been found only in the alpine zones of Mt. Katahdin, Maine; the Presidential range (mainly Mt. Washington), New Hampshire; and Mt. Marcy, New York; and on two cliffs in the Driftless Area of southwestern and south-central Wisconsin. The two Wisconsin sites are spectacularly disjunct from either the eastern mountain locations or the most southerly arctic stations along the southern shores of Hudson-James Bays, being removed 1250 km from both locations. At latitude 43°37'N (longitudes 89°47' and 90°37'W) and at 260 m above mean sea level, the two Wisconsin locations of
R. lapponicum
are the furthest south, low altitude stations in North America, and among the furthest south in the world for this species.
One of the two Wisconsin locations, at the Dells of the Wisconsin River (Columbia County) (photo 3) has been known to botanists since before the turn of the century. The first recorded account of the plant at this site was given by True (1898), who implies that the discovery of Lapland rosebay at the Dells by him and Professor L. S. Cheney of the University of Wisconsin in 1897 was the first scientific recognition of this plant in the U.S. west of the Adirondack Mountains. Still, one has to wonder whether this showy species had not been discovered locally before this time, since the Wisconsin River was one of the main early transportation routes in the Midwest, and the area was photographed extensively by one of the state's earliest photographers, H. H. Bennet.
The second location in Wisconsin for the species was only discovered in 1976 (Read and Smith, 1978). This location is 70 km to the west of the Wisconsin Dells, on a north-facing sandstone cliff (photo 4) along the upper Kickapoo River (Vernon County), in the heart of the Driftless Area.
What could account for
R. lapponicum
occurring in such anomalous habitats so far removed geographically from the main range of the species? While it is not the purpose of this paper to elaborate on the origins of the Wisconsin populations, there are two equally interesting theories on the subject which are presented below.
One theory holds that the Wisconsin populations represent relics of the flora that existed in the Driftless Area during, or at least immediately after, the last glacial advance about 12,000 years ago. There is a plethora of literature (e.g., Braun, 1951 ; Gleason, 1922; Curtis, 1959) suggesting that currently ranging boreal-arctic species survived continental glaciation of the last ice age in small refugia beyond (or perhaps actually on) the southern extent of the ice front. After glacial retreat these boreal-arctic species rapidly re-colonized the barren till and were eventually displaced in the south by more temperate vegetation migrating north. Only in the most protected locations within the former refuge areas, where precise microclimatic conditions prevail, do these relic species still exist.
The discovery by Baker (1965) of
R. lapponicum
seeds in early post-glacial bog deposits in northeastern Minnesota is the only fossil evidence known strongly suggesting that the species once ranged farther south in the Midwest. This bog deposit is about 925 km south of
R. lapponicum
's current range and about 400 km north of the extant Wisconsin localities.
The competing theory to the relic explanation of
R. lapponicum
and other unusual species in the Driftless Area is one that suggests that at least some of these species established themselves in receptive habitats by means of long-distance dispersal of reproductive propagules during the many thousands of years the habitats have been available for colonization. In the case of Lapland rosebay, this argument also seems plausible, since the seeds appear to be dispersible by a couple mechanisms.
The seeds are very small and light, and while not as light as the dust-like seeds of many orchids, under certain conditions the seeds could be distantly disseminated through wind dispersal. A sample of ten
R. lapponicum
seeds from the Wisconsin Dells site weighed an average of 4.3 x 10
-5
grams each, in the same order of magnitude as the Malaysian
R. verticillatum
(2.8 x 10
-5
grams/seed), listed by Ridley (1930) as being probably wind-dispersed. It is possible, too, that the seeds are animal dispersed, given the observation of Holmboe (in Ridley 1930) that
R. lapponicum
capsules are eaten by pine grosbeaks. Since pine grosbeaks do winter in Wisconsin, and have been observed in the Kickapoo Valley Lapland rosebay location feeding and roosting in conifers (
Tsuga canadensis
,
Pinus strobus
,
Pinus resinosa
), it seems plausible that an avian vector could account for this rhododendron's occurrence on Driftless Area cliffs. However, the migratory rates and food retention time of grosbeaks critical to this discussion are not known, and the short retention time of food noted in other birds studied (Ridley 1930) seems a limitation to this theory. Ecto-transport by birds is also not likely, given the small, smooth character of the Lapland rosebay seed.
Whatever the true reason for the occurrence of
R. lapponicum
in the Wisconsin Driftless Area, the presence of this arctic-alpine species on two cliffs is extraordinary and is evidence of unique conditions apparently not existing elsewhere in the Midwest. The habitats of both stations in Wisconsin are described in the following section.
Ecology
Unlike the dominant arctic-alpine habitat for Lapland rosebay of fellfield tundra, the Wisconsin habitats are both precipitous cliffs directly over running water of two rivers, the Wisconsin (the main river draining the state) and the Kickapoo (a major tributary to the lower Wisconsin River). At both localities, most plants seem to be well above the highest flood levels reached by the rivers, thus benefiting from the high and fairly constant local relative humidity generated from the rivers' surface, while remaining at high enough elevations off the river so as not to be washed off the cliff faces during seasonably high water levels. The majority of plants at the Wisconsin Dells site are located about 4.5 km off the non-flooded river level, while at the Kickapoo River cliff the lowest plants are 2.5 m off the normal river level.
Both cliffs are composed of fairly impervious, but jointed Cambrian sandstone (Pottsdam formation at the Wisconsin Dells, Franconian formation at the Kickapoo River). The joints are important, for they provide fissures in the rock faces where there is enough soil accumulation for vascular plant establishment. In the case of Wisconsin Dells population, these joints also provide a passageway for the slow emanation of water from an apparent perched water table in the vicinity. On all visits to the Wisconsin Dells population, a slow dripping of water from the base of the plants into the river below was noted. At the Kickapoo River population, the plants are anchored in certain fissures of the cliff face, but there is no active water flow emanating from within, although such is the case on other nearby cliffs in the Kickapoo River Valley not possessing
R. lapponicum
.
The two cliffs in Wisconsin containing Lapland rosebay possess different exposures which may account for the differences in water relationships between the locations. At the Wisconsin Dells site the cliff exposure is to the west and is thus exposed to direct sunlight for most of the hottest afternoon hours. Cold ground water emanating through the root zone and onto the rhododendron plants may therefore provide a critical cooling of the plants during periods of intense and direct solar radiation. On the other hand, the cliff exposure at the Kickapoo River Valley site (where there is no visible seepage) the cliff exposure is to the north, and the Lapland rosebay population receives no direct solar radiation whatsoever and there is therefore much less evaporation off the cliff faces.
That the cliff exposure and water supply relationships are importantly related to the microclimatic requirements of
R. lapponicum
in the Wisconsin Driftless Area is suggested by the distribution of this species on their respective cliffs. At the Wisconsin Dells the Lapland rosebay population is restricted to a single horizontal band at the summit of the cliff, limited in extent by active water seepage (photo 3). Only a single plant, removed from the main population by approximately 7 5 meters, does not adhere to this observation. The distribution of the rhododendron population on the cliff at the Kickapoo River Valley site (photo 4) is much different, being found in distinct, small clumps (individual plants?) scattered over the 15.5 meter high cliff face, from as low as 2.5 m to as high as 12.0 m above the normal river level.
The different cliff exposures and physical characteristics also account for few and various associates. On the xeric Wisconsin Dells cliff, red pine (
Pious resinosa
), jack pine (
P. banksiana
), polypody fern (
Polypodium virginianum
), sand cress (
Arabis lyrata
) and white lettuce (
Prenanthes alba
) may all be found within three meters of
R. lapponicum
. At the Kickapoo River site there are no vascular plants growing in the small crevices in which rhododendron is found, but the following, generally mesophytic species, do occur on the same cliff as does the Lapland rosebay: eastern hemlock (
Tsuga canadensis
), yellow birch (
Betula alleghaniensis
), and white pine (
Pious strobus
) are found at the cliff summit and in larger crevices on the cliff face, while florist's fern (
Dryopteris intermedia
), bulblet fern (
Cystopteria bulbifera
), northern willow herb (
Epilobium adenocaulon
), northern bugle weed (
Lycopus uniflorus
), clearweed (
Pilea pumila
), birds-eye primrose (
Primula mistassinica
) and sullivantia (
Sullivantia renifolia
) are found on the lower portions of the cliff, mainly within three meters of the normal river surface. The last two mentioned species are especially noteworthy, being two other vascular plants of special interest in the Driftless Area. The former is a boreal disjunct from the Great Lakes shorelines northward, while the latter is a regional endemic with a center of distribution in the Driftless Area. It should also be noted that these two species in particular also occur on shaded, damp cliffs of the Wisconsin Dells within one kilometer of the
R. lapponicum
.
With the preponderance of conifers at the cliff summits of both Lapland rosebay sites, it should not be surprising that the soil in which this species grows is very acidic (pH of 4.3 at Wisconsin Dells, 3.7 at the Kickapoo River Valley). The parent soil material present in the cliff crevices is mostly organic material which has been caught as material falls from the cliff summit above. Other mineral analyses from the native sites are given in Table 1, and are compared with soil in which transplanted
R. lapponicum
from the Wisconsin Dells has been grown successfully since 1973 (see the following section on propagation experience). Comparison of substrate characteristics in which Lapland rosebay grows natively and in cultivation shows quite a range for both macro- and micro- nutrient limits (or tolerances) for this species.
Table 1. Analysis* of native and cultural soil in which R. lapponicum is growing in Wisconsin.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Wisconsin
Dells |
4.3 | 1.6 | 6.5 | 4.1 | 0.276 | 41.0 | 1.00 | 0.500 | 0.100 | 0.225 | 3690 | <20.0 | 1550 | <10.0 | 14.2 | 37.9 | 25.3 | 26.6 | <30.0 |
|
Kickapoo
Valley |
3.7 | 7.1 | 5.2 | 10.3 | 0.524 | 131.4 | 1.00 | 0.500 | 0.126 | 0.346 | 11800 | 133.0 | 5800 | 12.1 | 42.6 | 24.0 | 40.5 | 58.6 | 34.3 |
|
Culture soil
(Wrzesinsk) transplants from Wis- consin Dells 1973) |
6.9 | 12.2 | -- | 61.0 | 1.75 | 61.0 | 1.07 | 2.07 | 0.670 | 0.472 | 13700 | 207 | 22800 | 59.8 | 116 | 30.9 | 50.9 | 386 | 35.3 |
*Conducted by Soil and Plant Analysis Laboratory, University of Wisconsin-Extension.
**Elemental analysis by Emission Spectroscopy (HF treatment).
No critical morphologic comparisons have been made between plants of the main arctic range and those of the isolated populations on mountains in New York and New England, and on cliffs in Wisconsin. If these latter outlying locations have been isolated for thousands of years as some believe, divergence of certain characters may have occurred. But pending such a critical evaluation of the species, we have observed differences in flowering dates and general stature in the Wisconsin populations from the main range and eastern mountain habitats that are suggestive of fixed variation. The Wisconsin plants appear to be consistently more robust than arctic-alpine plants, the former being often over 40 cm tall as compared to the 10-30 cm tall figures given for the species in eastern North America.
|
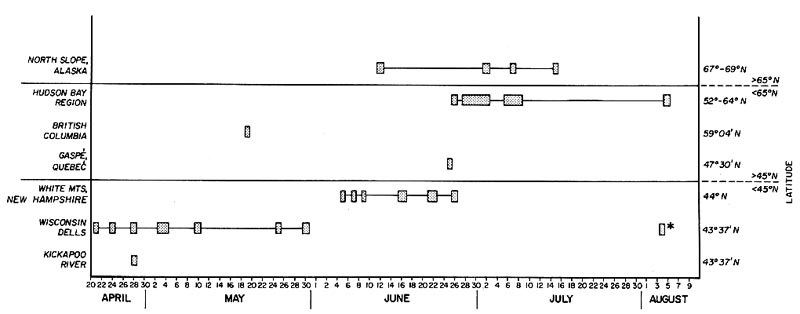
Figure 2. Range of flowering dates for
R. lapponicum
from different locations shows that plants in the Wisconsin cliff habitats consistently bloom earlier than those in arctic alpine tundra habitats of higher latitude or altitude. Whether this is due to ecotypic differences or simply to latitude-altitude differences is not known. the late blooming date noted in the Wisconsin Dells population (*) is a weak second flowering of certain plants triggered by the long summer season and decreasing day length. Flowering dates were gathered from personal observations (Wisconsin populations) and collections in the herbaria of the University of Wisconsin-Madison, University of Wisconsin-Milwaukee, Milwaukee Public Museum and the University of Minnesota.
Another observation in the Wisconsin plants that is suggestive of ecotypic variation is that of flowering dates. Figure 2 shows that Wisconsin
R. lapponicum
almost always blooms earlier than plants of arctic or alpine habitat - by as little as one week, but often by more than one month. Plants at the Wisconsin Dells colony have also been noted to undergo a second, but limited, blooming period in mid-August, probably triggered by the long summer season and decreasing day length. Interestingly, the spring blooming dates for
R. lapponicum
growing at the Wisconsin Dells corresponds here in USDA plant zone 5a with the widely planted rhododendron selection 'PJM' (
R. dauricum
x
R. carolinianum
).
The distinctly earlier blooming period of R. lapponicum in Wisconsin, as well as the seemingly larger stature of these same plants, may only be due to the unique environmental conditions and low latitude /elevation of the Wisconsin cliff habitats, but it is tantalizing to think that genetically fixed morphologic, phenologic, and ecologic adaptations may have developed in these highly isolated populations. Further studies with these interesting Wisconsin colonies are needed to answer these many speculations.
Rhododendron lapponicum as a Cultivated Plant
As mentioned previously,
R. lapponicum
is one of the most widely naturally distributed rhododendrons in the world. Its floral beauty and low growth form bestow this species with exceptional horticultural potential. Yet,
R. lapponicum
has proved difficult to establish in cultivation. Usually plants collected in the wild soon die or exist in a weakened condition for a year or two and then succumb. Most of the plants of
R. lapponicum
used in propagation experiments have come from sources in Canada, Alaska or Mt. Washington, New Hampshire. Through personal contacts, correspondence and a search of the literature, no mention was found of anyone that has worked with plants of
R. lapponicum
from the alpine zones of Mt. Marcy in Essex County, New York or from Mt. Katahdin in Maine.
A review of some of the recent experiments with Lapland rosebay of arctic and alpine origin will illustrate the chronic problems encountered in establishing this species in a garden situation. The Montreal Botanic Garden in Quebec has several plants grown from seed collected two years ago on islands in James Bay in the western part of Quebec Province. The Royal Botanical Gardens in Hamilton, Ontario, has a plant of
R. lapponicum
in their collection. This plant was obtained from the Ontario Horticultural Research Institute in Vineland Station, Ontario in 1960. The plant remained in a lath house until 1969, when it was set out in the Garden's Rhododendron collection. According to Freek Vrugtman, Curator of Collections of the Royal Botanical Gardens, their specimen has not been propagated further.
R. lapponicum
is no longer in the plant collections of the Horticultural Research Station at Vineland Station, Ontario. Attempts to re-establish the plant from seed and from plants collected in the Arctic have not been successful.
According to D. S. Craig of the Canadian Research Station at Kentville, Nova Scotia,
R. lapponicum
has been in their collection since 1956. The plant was grown from seed collected on Mt. Washington, New Hampshire. (This plant was featured on the cover of the January, 1967 issue of the "Bulletin".) The plant began to deteriorate in the late 1960's. Cuttings taken from this original plant rooted easily but are not really vigorous plants. Mr. Craig speculates that a lower soil pH might make a difference with the plants of
R. lapponicum
at Kentville.
The University of Washington Arboretum in Seattle has tried to establish the plant several times over the years without success. The Arnold Arboretum has tried
R. lapponicum
from both cuttings and seed collected at Mt. Washington in New Hampshire without success. According to Gary L. Koller, Supervisor of the Living Collections, the Arnold Arboretum currently has six seedlings (371-73) 10-15 cm tall in their collection. These plants of
R. lapponicum
were grown from seed obtained through the American Rock Garden Society at Mile 400 of the Alaska Highway. The Arnold Arboretum has not tried any plants from the colony of
R. lapponicum
on Mt. Marcy in Essex County, New York. The University of New Hampshire at Durham does not have any plants of
R. lapponicum
in their collections at the present time.
Several plants of
R. lapponicum
collected along the shores of Great Slave Lake in Canada are doing fairly well at the Royal Botanic Garden in Edinburgh, Scotland. Attempts to establish
R. lapponicum
at the Royal Botanic Garden from other sources have not been successful. Currently a batch of seed from Oulu in Finland have germinated, but according to Dr. James Cullen of the Royal Botanic Garden, these seedlings are extremely slow growing. From time to time plants of
R. lapponicum
have been given to the Crown Estate Gardens at the Great Park in Windsor, England. It has proved difficult to keep these plants at the Crown Estate Gardens alive for any length of time.
Besides the experience of these institutions, several individuals have also had varying degrees of success with
R. lapponicum
in cultivation. Dr. J. B. Brueckner of Mississauga, Ontario is growing an upright form of
R. lapponicum
that was collected along the shores of Great Slave Lake in Canada. Peter Cox of Perth, Scotland, is also growing
R. lapponicum
from this same source. Fred Case II of Saginaw, Michigan, is growing a mat forming type from Mt. Washington and another form from seed collected on Mt. McKinley in Alaska.
To our knowledge, no propagation research has been directed to Lapland rosebay from the native Wisconsin sites. In the hope that these plants of low latitude, low elevation, isolated distribution, and anomalous habitat might possess characteristics more conducive to cultivation the junior author initiated propagation experiments with the Wisconsin Dells plants in 1974. The junior author has been cultivating rhododendrons as an avocation since 1972 and has over 100 species and cultivars from all over the world in his garden in Middleton, Wisconsin, just west of Madison (USDA Zone 5a). His efforts represent the largest rhododendron collection in Wisconsin.
Two small plants with roots were collected from the edge of the small Wisconsin Dells colony for garden trial on 1 April 1974. Since there was concern that the plants might not survive the move, larger budded plants were not collected. It was recognized that this unique colony should not be depleted by the removal of larger budded plants. The two small plants were set out in the junior author's rock garden in Middleton, Wisconsin (64 km to the south) later that day.
Both of the Wisconsin Dells transplants survived the move. Their garden location faces east, which provides late afternoon protection from the sun in the shade of the author's house. The soil is well drained sandy loam (see Table 1). The plants do require an ample supply of moisture. Supplemental water during the summer has been applied. In coming years after the plants are more established, the supplemental water hopefully will not be necessary. Further study of moisture requirements are needed.
Summer heat has not been a problem with the plants of Lapland rosebay collected from along the Wisconsin River. The two plants have been well mulched with white pine (
Pious strobus
) needles. This mulch is renewed periodically.
New growth has been observed during each succeeding year following the transplant. The growth rate is difficult to determine until more time has elapsed, but during the summer of 1977 about 2-3 cm of growth was observed. Much of this growth was lateral branching. This growth habit should produce a plant with a low mounded appearance in coming years. While there has been no flowering of the transplants to date, one of the plants set buds for the first time during the late summer of 1977. This plant is branched with a maximum vertical height of about 15 cm.
In the junior author's rhododendron collection is another plant of
R. lapponicum
, a seedling of the form collected near Great Slave Lake in Canada. This plant was received in September, 1975, and while not enough time has passed for an accurate comparison, the Great Slave Lake form does seem more sensitive to summer heat. The plant is also not as vigorous as the plants collected from the colony along the Wisconsin River. During the summer of 1977 this form of
R. lapponicum
grew less than one centimeter. This limited growth was also of a lateral nature. In late August of 1977, this Great Slave Lake form was moved to a more shaded location. This move may have a beneficial effect on the growth of this plant. Evaluation of this form of
R. lapponicum
needs to continue before any substantive comparison can be made. Some rhododendrons need to be established for several years before commencing a normal pattern of growth.
In the fall of 1976, some cuttings of
R. lapponicum
from the Wisconsin Dells colony were sent to Mrs. Lucie Sorenson of the Bovee Nursery in Portland, Oregon. Mrs. Sorenson has reported that the cuttings rooted and grew so well that second cuttings have been taken from the new growth.
The final effort that is being made to propagate
R. lapponicum
of Wisconsin origin is by seed germination. In October, 1976 an attempt was made to collect seed from the Wisconsin Dells colony. This attempt was disappointing; the capsules had opened completely and dropped their contents into the water below. The following year an earlier attempt was made on 4 August 1977. Even by August most of the capsules had opened.
Fortunately some seed was collected from the remaining capsules. This seed was divided into ten lots of 100 seeds per lot. No significant amount of seed has been collected from the Kickapoo River Valley colony.
Several of these lots are under germination trials here in the winter and spring of 1978. The rest have been distributed for propagation and trial under growing conditions elsewhere. This distribution of seed will provide, if successfully propagated, valuable information on the adaptability of this form of
R. lapponicum
to various growing conditions.
People participating in this distribution include: Mr. Harold Hillier, Hillier Nurseries, Winchester. England; Tor Nitzelius, Vaxtorp, Sweden; Fred Case, Saginaw, Michigan; Jim Gerdemann and Don Paden, Urbana, Illinois. Seed was also made available to Dr. Melva N. Philipson, Christchurch, New Zealand.
In the spring of 1978, an attempt will be made to collect pollen from the plants in the colony along the Wisconsin River. Several individuals have expressed an interest in cooperating in the possible hybridizing of this form of
R lapponicum
. Hardiness, possible heat tolerance, and a dwarf compact habit are some of the traits that might be imparted from this form of
R lapponicum
. More information of this effort will be reported later.
Acknowledgements
The authors wish to thank the many correspondents around the world who offered to share their experience in propagating
R. lapponicum
. Appreciation is extended to the curators and directors of the herbaria of the University of Minnesota, University of Wisconsin-Madison, University of Wisconsin-Milwaukee, and the Milwaukee Public Museum for access to their
R. lapponicum
collections. Part of the field investigations for this work were supported through a contract with the U.S. Army Corps of Engineers.
Literature Cited
Baker, R. G., 1965, Later glacial pollen and macrofossils from Spider Creek, southern St. Loins County, Minn. Bull. Geol. Soc. Amer. 76:601-610
Braun, E. L., 1951, Plant distribution in relation to the glacial boundary, Ohio J. Sci., 51.139-146
Curtis, J. T., 1959, The Vegetation of Wisconsin, Univ. Wis. Press, Madison, 657 pp.
Gleason, H A., 1922, The vegetational history of the Middle West, Ann. Assoc. Am. Geog, 12:39-85.
Hulten E., 1958, The Amphi-Atlantic Plants and Their Phytogeographical Connections, Sv. Vet Acad. Handl. Bd. 7 Nr 1 Almquist and Wiksell, Stockholm, 340 pp
Philipson, M. N. and W R Philipson, 1975, A revision of
Rhododendron
section Lapponicum, Notes Royal Bot. Gard., Edinburgh, 34 1-71
Read, R H. and W. E. Smith, 1978, In Prep. A new location for
Rhododendron lapponicum
in Wisconsin's Driftless Area
Ridley, H. N., 1930. The dispersal of plants throughout the world, Reeve Press, Ashford
True, R. H., 1898, Botanizing in the Dalles of the Wisconsin River, Plant World, 1(6):81-83.