Insecticidal Soap As An Azalea Lacebug Control
Stanton A. Gill & Michael J. Raupp
University of Maryland
Ellicott City, Maryland
Introduction
Insecticidal soaps are increasingly marketed as safe, effective materials for suppressing insects and mites. Soaps exhibit relative selectivity in the range of pests affected. They are also relatively non-persistent in the environment. These benefits combined with their low mammalian toxicity make soaps a promising tactic for use in integrated pest management programs.
Soaps have been used for insect suppression since the later half of the nineteenth century. The mode of action and efficacy of various soaps was described before 1940 (Van der Muelen and Van Leeuwen 1929, Fulton 1930, Dills and Menusan 1935). Insecticidal soaps, such as Safer Soap, are composed of potassium salts of several fatty acids. The mode of action of these fatty acids is believed to disrupt the pest's cellular membrane which results in loss of cellular contents, thus causing the cells to die (Osborne and Henley 1982). Little published work was done with soaps during the period of 1940-1970 (Osborne 1984). Some of the early research into modern-day insecticidal soaps was initiated by George Puritch of Pacific Forest Research Center at the Canadian Forestry Service (Pinnock et al. 1974).
Koehler and his co-workers (1983) used insecticidal soaps to effectively suppress whitefly and cabbage worms. Osborne (1984) of the University of Florida Foliage Research Center reported good spider mite control on schefflera in greenhouse tests. Osborne indicated that 6.2% or 12.4% A.I. of soap provided control as effective as 0.3% dienochlor (PENTAC). He also noted that soap residues remained effective against
Tetranychus urticae
at least twenty-four hours after treatment. He suggested that the effect might be repellency rather than toxicity since no dead mites were observed.
The azalea lacebug,
Stephanitis phyrioides
(Scott), is one of the top ten most common insect pests in Maryland landscapes (Holmes and Davidson 1984, Raupp and Noland 1984). Safer Agro-Chem's insecticidal soap has been registered in the U.S. as an insecticide. It is labeled for control of several soft-bodied arthropod pests on a wide range of landscape ornamentals. To investigate the efficacy of insecticidal soap as a lacebug suppressor, the following experiment was conducted.
Materials and Methods
Twenty evergreen azaleas in a mulched hardwood bed were selected at Columbia College, Takoma Park, Maryland. The bed contained equal numbers of the cultivars 'Hershey's Red' and 'Delaware Valley White', 2.5 to 3.0 feet high and 3 to 4 feet wide. Plants were in full sun from approximately 10 am until 5 pm during the experiment in May. The plants were separated by sidewalks into four blocks of five plants. Plants in the first bed remained untreated during the course of the study. The second group of five plants was treated with a single spray of insecticidal soap at a rate of 5 tablespoons/gal. The spray was applied so that undersides of foliage were treated. A five gallon Birschmeyer sprayer with a hollow cone spray nozzle was used. The third group of five plants was treated with acephate (Orthene) at a rate of 0.125% A.I. The last block of five plants received insecticidal soap at the rate previously described for the second group.
Prior to the treatments, lacebug densities were determined by sampling each plant. The samples consisted of removing 5" from ten terminals on each plant. The number of lacebug nymphs found on each terminal was recorded.
Forty-eight hours after the treatments, ten samples of 5" tip growth were taken randomly from each plant. The number of live lacebug nymphs was recorded for each sample. Vitality was determined by probing lacebugs with a needle several times. If there was no response, then death was assumed. Statistical analyses were performed on the number of living nymphs found in each sample.
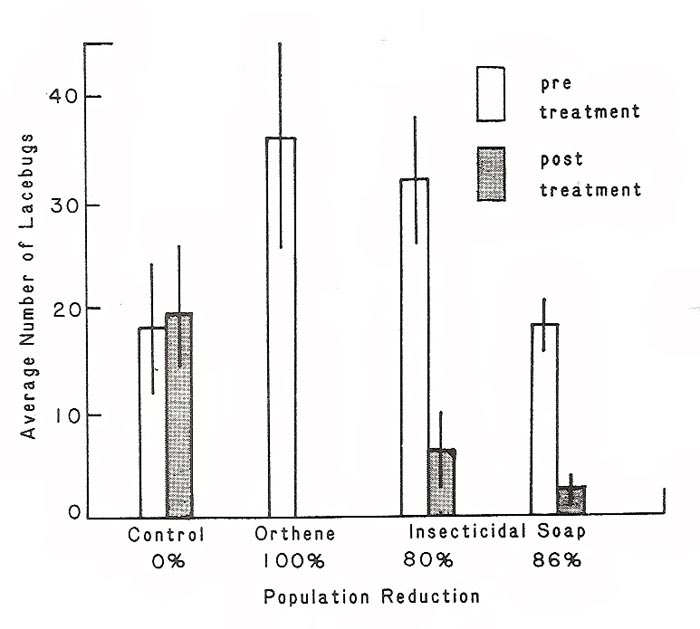
|
|
Figure 1. Number of lacebugs per five inch shoot of azalea, prior to and
two days after insecticide treatments. Bars represent averages of 5 plants, vertical lines represent standard errors. |
Results and Discussion
The following analyses were performed. First, to be sure that no bias had been introduced into the experiment, the densities of lacebugs on the plants prior to treatments were compared with the Krusakal-Wallis oneway analyses of variance. No significant differences in lacebug densities were observed among treatments prior to the application of insecticides (x
2
= 5.06, p < 0.167).
However, following the applications, lacebug densities differed significantly among treatments (x
2
= 14.25, p < 0.003). The untreated control plants experienced no change in lacebug numbers two days after the treatment (Figure 1). Lacebugs on azaleas treated with acephate were all killed, Insecticidal soaps were also extremely effective in reducing lacebug abundance. In one block lacebugs were reduced by 80% and in the other an 86% reduction was observed (Figure 1).
These results clearly demonstrate the efficacy of two insecticides commonly recommended for lacebug control. Acephate has a slightly higher kill rate than insecticidal soap. However, insecticidal soap has an extremely low mammalian toxicity. Both materials provided good control of the azalea lacebug and are acceptable tactics for use in integrated pest management programs for ornamental plants. No injury to the azaleas was noted.
This spring, a follow-up experiment will test the efficacy of horticultural oil (2%) and a lesser rate of insecticidal soap spray for controlling lacebug on evergreen azaleas.
Literature Cited
1. Dills, L.E., and H. Menusan. 1935. A study of some fatty acids and their soaps as contact insecticides. Contribution Boyce Thompson Inst. 7:63-82.
2. Fulton, B.B. 1930. The relation of evaporation to killing efficacy of soap solutions on the Harlequin Bug and other insects.
J. Econ. Entomol.
23: 625-630.
3. Holmes, J.J. and J.A. Davidson. 1984. Integrated pest management for arborists: Implementation of a pilot program in Maryland.
J. Arboric.
10:65-70.
4. Koehler, C.S., L.W. Barclay, and T.M. Kretchun. 1983. Soaps as pesticides.
California Agriculture
, Sept-Oct. pp. 11-12.
5. Osborne, L.S. 1984. Soap spray: An alternative to a conventional acaricide for controlling the two-spotted spider mite (Acari: Tetranychidae) in greenhouses.
J. Econ. Entomol.
77:734-737.
6. Osborne, L.S. and R.W. Henley. 1982. Evaluation of Safer Agro-Chem's insecticidal soap for the control of mites in the interior environment. ARC - A Research Report RH-82-2 Agriculture Research Center - Apopka, FL.
7. Pinnock, D.E., R.J. Brand, J.E. Milstead, and N.F. Coe. 1974. Suppression of populations of
Aphis gossypii
and
A. spiraecola
by soap sprays,
J. Econ. Entomol.
67:783-784.
8. Raupp, J.J. and R.M. Noland. 1984. Implementing landscape plant management programs in institutional and residential settings.
J. Arboric.
10:161-169.
9. Van der Meulen, P.A., and E.P. Van Leeuwen. 1929. A study of the insecticidal properties of soap against the Japanese Beetle.
J. Econ. Entomol.
22:812-814.
Stanton Gill is Regional Agent, Maryland Cooperative Extension Service, Clarksville Experimental Farm, Ellicott City, Maryland.
Michael Rauff is Extension Specialist, Department of Entomology, University of Maryland, College Park, Maryland.