Causes of Brilliant Color in the Flowers of Rhododendron 'Creole Belle'
Woodard Ching
Bellingham, Washington
In the mid-1990s I noticed a stunning rhododendron with light purplish red flowers. It was 'Creole Belle'. The experience started me wondering why the flowers of this plant appeared so brilliant. Then, in 1999, I read an article in the Journal American Rhododendron Society by Donald Hardgrove; he had also been struck by the brilliance of the flowers on a rhododendron - 'Glowlight'*. His observations piqued my interest even more:
What caused this glow, a phenomenon that I would later come to call "brilliance"? The brilliance of 'Creole Belle' seemed so obvious to me that I pondered how the plant achieved it (Fig. 2). The plant, an elepidote rhododendron from a cross of 'Vulcan' x 'Harvest Moon', was introduced by W. Thompson in 1982 (12) and its name was registered in 1990 (see below) (13). My curiosity about the cause of the glowing color in its flowers prompted me to begin an investigation that, over the next three years, would draw me into various disciplines including biology, psychology, physics, chemistry, and art.

|
|
Figure 2. Flower of 'Creole Belle'.
Photo by Woodard Ching |
Color - A Human Perception
The color appearance of an object is a human perception (7). It results from interaction of four elements: a light source, an object, the human eye (receiving light reflected from the object and generating certain neural signals), and the human brain (interpreting the neural signals transmitted from the eye). The human visual system's functioning above very dim light levels involves excitation of the color-sensitive cones in the retina of the eye. At very low light levels, the cones do not respond, and non-color-sensitive rods send signals that the brain interprets only as white, gray, and black (8).
Before turning to the perception of color in plant tissue, consider the color of a red barn. Rays of light from the sun and sky (the source) contain wavelengths of light from red to violet: this may be seen in a rainbow or by passing the light through a prism. (Purple is not found in the spectrum; it comprises red and blue wavelengths) (7). When light from the source strikes the barn (the object), a few rays reflected from the painted surface may retain their full (or nearly full) spectral qualities. Most will contact the pigment of the paint, which will absorb green and blue wavelengths and reflect only rays that will be interpreted by the eye and brain as "red."
Unlike the barn, living tissue is not opaque. To provide a basis for describing the paths and wavelength changes of light rays striking the petal of a flower, we must first consider some details of cell and tissue structure (3). Typically, an elepidote rhododendron petal has an upper epidermal layer; a sub-epidermal layer that may be more than one cell in depth; a spongy mesophyll with irregular lacunae (air spaces) between the living cells; a lower sub-epidermal layer; and a lower epidermis. The outer surface of the petal may be coated with a waxy substance (cutin). While the cells comprising these layers may differ in size, shape, and presence of pigmentation, they share many features in common. The structure of individual cells, as well as their composition in the layers of the petal, affects perceived color (19).
Basic to the structure of a cell is a more or less rigid outer cell wall composed largely of cellulose (Fig. 1) (3). Inside the wall, a cytoplasmic membrane composed of protein and fatty substances encloses the living part of the cell, the protoplasm. The protoplasm includes the nucleus, containing the cell's genetic material, and the cytoplasm in which metabolic functions occur. In the cytoplasm - a semi-fluid or jellylike substance - are found a variety of "organelles" of various sizes. Among these are the chloroplasts containing chlorophyll, the key to photosynthesis and the source of green color in plants, and chromoplasts that may carry yellow and orange carotenoid pigments. And, perhaps most significant in considering the color of 'Creole Belle', the interior of the cell includes a large sac - the vacuole - that contains much of the cell's stored water, excess mineral nutrients, and soluble flavonoid pigments such as anthocyanins and flavonol co-pigments. Adjacent cells are bound together by a thin layer of pectin.
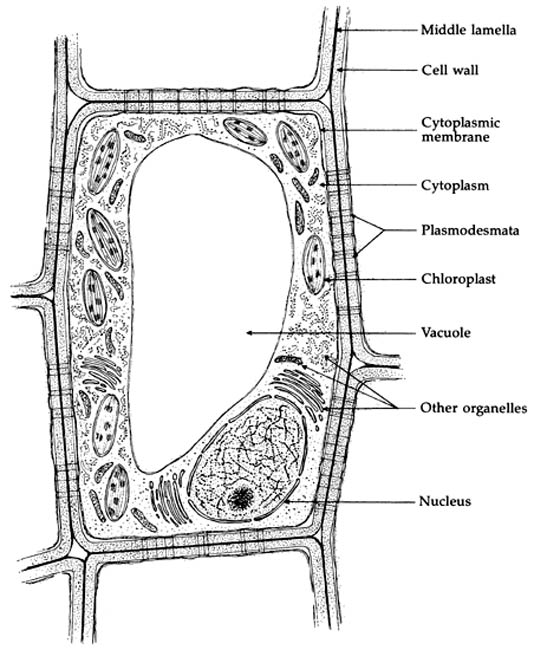
|
|
Figure 1. Details of a plant cell.
Drawing by Brian Capon In his book Botany for Gardeners published by Timber Press, Portland, Oregon. |
Why consider all of this detail? Rays of light falling on a petal are subject to reflection, transmission, absorption, refraction, and scatter (8). When light rays are incident to the petal surface at an angle other than exactly perpendicular, at least a small part of the light is reflected and, as the rays penetrate below the surface, refraction alters their direction. Change in direction and slight reflection occur at each interface between materials with different refractive indices. Each additional encounter with cell walls and various cell contents produces reflection, refraction, scatter, and transmission. Pigments and - to a minor degree - other cell substances absorb certain wavelengths of light. These actions recur until the light that entered the petal tissue has been reflected back through the illuminated side of the petal (often changed in spectral quality), absorbed within the petal, or transmitted through the petal.
Color appearance is also affected by surface characteristics such as gloss and texture (7) and by the nature of the incident light (strongly directional, as sunlight, or diffuse). A smooth surface will produce some specular (mirror-like) reflection, while a textured epidermis will result in diffuse reflection and a greater proportion of light rays entering the petal. The spectral composition of the light rays that are finally reflected toward the observer thus depends on a variety of factors (3). Some rays may be reflected from the surface with little or no spectral absorption and thus trigger neural signals coded for "white." Other rays, having penetrated the petal and been subjected to absorption of various wavelengths of light, primarily by contact with the pigments, may reach the observer's eye and trigger neural signals for a color such as "red." Then, depending on the relative number of rays coded for red and for white in the light reaching the eye, the brain will integrate the signals and produce a sensation of lighter red or pink. Surface characteristics may also affect the perception of color (8).
A Special Case - Twilight to Dark
I believe in experiments that people can do without needing a biology laboratory, microscopes, etc. One evening in 1999, I gathered blossoms that seemed to glow; these were selected from a number of rhododendron hybrids that people describe as "hot" colored. I placed them in a row on a tree stump near my house and sat on a lawn chair for nearly two hours to observe them as the sun set and darkness fell. There were ten blossoms: 'Atroflow', 'Lydia', 'Hallelujah', 'Isabel Pierce', 'El Camino'*, 'Hydon Dawn', 'Ross Maud', 'The Honourable Jean Marie de Montague', 'Burnaby Centennial', and 'Creole Belle'. I observed that, one by one, all of these blooms gradually came to appear black until only two still showed color: 'Burnaby Centennial' and 'Creole Belle'. In the case of 'Creole Belle', the purplish petal color appeared to become more bluish.
As the setting sun nears the horizon, the color of ambient light reddens. Surfaces perceived as white in full daylight now reflect orange light. But through a phenomenon known as "memory color" (7, 9) the brain shifts the perceived color toward white. As the sunset proceeds, the red fades and the blue light of the sky predominates. From the time when the setting sun is a few degrees below the horizon to the time when night is illuminated by no more than a half-moon, both cones and rods are active in the retina of the eye, permitting some perception of color.
As near darkness approaches, cone vision remains active for a time, but the color sensitivity of the eye shifts toward the blue end of the spectrum. This shift is known as the Purkinje effect. As illumination decreases, a point is reached at which the appearances of red and blue objects that had been perceived as equal in lightness will change. The lightness of the red object will now be seen to decrease in relation to that of the blue object. With a further decrease in the ambient light, cone response ceases; only rod vision is active. Vision is now achromatic: the red object appears black and the blue object, gray (sometimes a bluish tinge persists for a time) (7).
These effects undoubtedly contributed to the visibility of 'Creole Belle' flowers after most of the others in the tree-stump experiment came to appear black. The blue-wavelength component of the purplish coloration in 'Creole Belle' would remain visible as gray until darkness extinguishes discrimination.

|
|
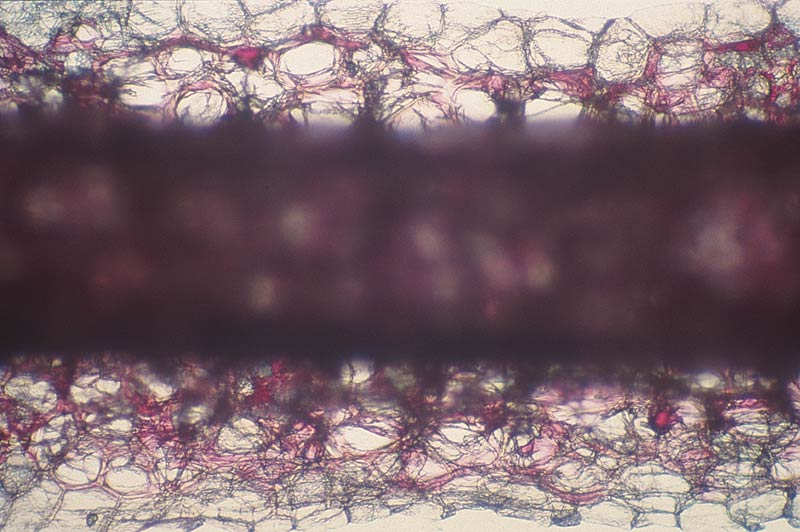
Figure 3. Cross section of a typical red-flowered elepidote rhododendron petal.
Note the outer layers of red-pigmented cells and the inner layer of white cells. Photo by Woodard Ching |
Into the Laboratory
Fascinated by the brilliant appearance of 'Creole Belle' and curious to seek further explanation for the outcome of my "tree stump" experiment, I took a course in plant cell biology and found a biologist who permitted me the use of her laboratory and a light microscope. Before my 'Creole Belle' bloomed, I obtained rhododendron blossoms from gardens of members of my ARS chapter, Komo Kulshan.
The procedure I developed was to cut very thin cross-sections from single petals, pulling a sharp razor blade from the center toward the margin. The sections were mounted in de-ionized water on glass slides. Although I learned much from study of these sections, for many reasons I recommend that anyone requiring slides of plant tissue for research obtain the services of someone skilled in the techniques of slide preparation.
By the time my 'Creole Belle' bloomed, I had examined a number of early blooming red rhododendrons, all believed to be bright in color. Upon examination under magnification, the petals all had layers of colored cells. The usual order was an upper epidermis of red-pigmented cells, below it a layer of colorless cells, and below that the lower epidermis of red-pigmented cells (Fig. 3). (Petal tissue with cells that are colorless when viewed by transmitted light under the microscope may appear white when viewed by reflected light.)
When the first slides of 'Creole Belle' came into focus, I was astonished. It did not look at all like 'Taurus' and 'The Honourable Jean Marie de Montague'! Near the base of the petal, the epidermal layer was a very saturated deep purplish red, below it was a thick middle layer of colorless cells, and below that another layer of deep purplish red cells. Each of the three layers was three cells thick. A few millimeters toward the apex from the dark center, the order of the cell layers was reversed: the outer layers were clear and the inner layer was the deep purplish red (Fig. 4).
|
|
Figure 4. In 'Creole Belle' the outer layers of cells are white and cells in
the middle layer of the petal contain a very deep purplish red pigment. Photo by Woodard Ching |
The distribution of pigmented cells in 'Creole Belle' petals revealed by my cross sections raised a plethora of questions.
• Do the lacunae (air spaces between cells) in the spongy mesophyll have a special function in reflecting light?
• Lightness in a color may contribute to brilliance, but the described color of 'Creole Belle' corresponds in lightness to a middle gray, and many other purplish red rhododendrons of approximately the same lightness are not considered "brilliant." What role does the layer of white cells play?
• Why is the described color of the flare in the throat of 'Creole Belle' a deep red, not the strong purplish red of the outer portions of the corolla or the deeper pigment color observed in cross sections?
• Are factors outside the composition of the corolla responsible for the observed brilliance of 'Creole Belle'?
A search for evidence of white pigmentation in the flowers of 'Creole Belle'; examination of the ways in which light is reflected, scattered, and absorbed in petal tissue; and the relation of observed pigment color within a petal and the color appearance of the petal would be the focus of my research in the next two years.
2001 - 2002
In 2001, my attention turned to investigating whether there are extremely small white solids (organic or inorganic) in the cells of 'Creole Belle' that reflect sunlight. The possibilities included white or near-white pigments, starches, and crystals. Other explanations considered for a flower's appearing white were surface conformation (wet or dry surface conditions), co-pigmentation, pH changes, and lacunae (air spaces between cells).
Calcium oxylate crystals, known to occur in the flowers of certain taxa (Poinsettia is the best example), would not only reflect nearly full-spectrum (white) light rays but also contribute to the appearance of brilliance. These crystals may be present in two forms, druses (ca. 5µm in size) and raphides (ca. 50µm in size) (16); when present they can be seen with a light microscope. Their appearance resembles that of gold dust in a mountain stream but is often referred to as "diamond dust." These crystals were not found in any of the large number of flowers from various elepidote rhododendrons cultivars that I examined.
The question of how the appearance of white is produced by cells without the presence of visually observable white pigments was a subject for investigation in 2002. This involved detailed examination of petal tissues and consideration of the paths taken by light rays that strike a petal. Cell morphology and the principles of geometry affecting reflection, refraction, scatter, and transmission of light were studied.
When a ray of sunlight falls on a petal, the complex repetitive processes of reflection, refraction, scatter, absorption, and transmission occur as described earlier. Of special interest to discussion of the color appearance of 'Creole Belle' is the way that absorption alters the spectral (wavelength) properties of light rays.
With respect to the appearance of whiteness, Swain (20) (an expert on flavonoid compounds) has stated: "Yet other flavonoids account for the actual whiteness in most white flowers, which without them would perhaps appear almost translucent." These co-pigments have absorption properties that usually yield yellow or greenish yellow coloration - but at such low chroma that the flowers are seen as white. When viewed through the microscope with transmitted light, the cells containing these co-pigments may appear clear or cloudy.
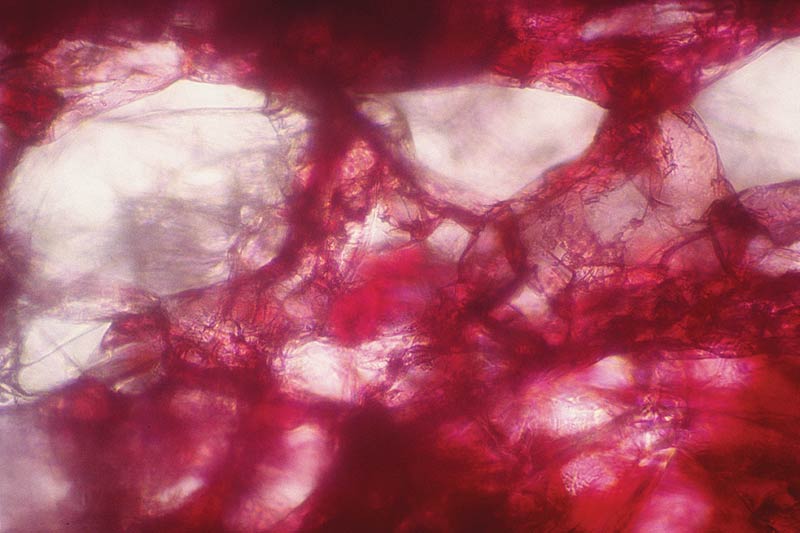
An interesting situation occurs when an interior cell wall is adjacent to an air space rather than another cell wall (Fig. 5). Spethmann (19) has stated that the white appearance of a flower can result from "reflection of the visible light by the air enclosed in the mesophyll"; Percherer (15) held that the white color "is due to the air-filled lacunae, i.e., empty cells." (Note, however, that the lacunae are air spaces between cells, not empty cells). But light travels through air, and air alone does not reflect light.
|
|
Figure 5. Cross section of petal showing lacunae (large air spaces) in the spongy mesophyll tissue.
Photo by Woodard Ching |
A tested explanation for the contribution of lacunae in petal mesophyll to the reflection of light rays was not found in my search of the literature. Unlike pure dry air, however, the air in lacunae within the petal probably contains water vapor or minute droplets. Thus one may hypothesize that, at a certain range of particle sizes, an incident beam of white light will scatter strongly. In the absence of change in the spectral quality of the light due to absorption, the scatter would create the appearance of white in the lacunae. The relative contribution of this to the perceived whiteness of a petal also depends on the amount of reflection from the petal surface and from cells at various levels; light incident on the lacunae and reflected from them must traverse a layer of tissue that may be several cells thick (2).
The red and purple coloration in rhododendrons results from a major change in the spectral quality of a light ray occurring in a petal cell vacuole containing flavonoid pigments and co-pigments (such as anthocyanin and flavonols). In general, the effect of anthocyanin is to produce red and, combined with flavonol co-pigmentation, purplish red. Other color-changing absorption may occur in a flower if a light ray encounters chromoplasts (producing orange and yellow) and chloroplasts (producing green) in the cytoplasm.
The registration description of 'Creole Belle' lists the corolla color as strong purplish red (RHS 64C) with deep red (RHS 53A) (17) in the throat. One explanation for the strong purplish red color seemed evident. Rays of sunlight pass through the epidermis and sub-epidermal cells in the top layer (which by itself appears white) to the deep purplish red cells of the middle layer. Spectral absorption occurs here, and the light reflected back through the upper layer emerges from the petal largely unaffected in spectral quality by the cells of the upper layer. These rays now carry the red and blue wavelengths that combine to produce purplish red; together with the nearly full spectrum ("white") rays reflected from the upper layer of the petal, they produce in the human visual system a purplish red that is lighter than the pigment color observed during examination of the cross sections.
The contrast perceived between the strong purplish red of the corolla with the deep red flare in the throat may be enhanced by a perceptual phenomenon known as simultaneous contrast (6). Especially where the two colors abut, the darker color will appear even darker; the lighter color, even lighter. This contrast may be a factor contributing to the observed brilliance of the flowers of 'Creole Belle'.
Speculations and Issues for Future Research
Two photographs reproduced in Salley & Greer's Rhododendron Hybrids (18) suggest hypotheses concerning the brilliance question and the red hue of the flare at the base of the 'Creole Belle' corolla. The photograph of 'Creole Belle' by Harold Greer (Plate 115) provides clues relating to the appearance of brilliance. The upper (adaxial) petal surfaces not illuminated by direct sunlight, as well as lower (abaxial) surfaces illuminated by transmitted light, appear to be a fairly even purplish red. But, depending on the angles of incidence and reflection of light, parts of the petals illuminated by direct sunlight show a prominent whitish sheen. A relatively smooth epidermis covered with a waxy cutin would contribute to this appearance. The Greer photograph suggests an additional reason for the brilliance noted in blossoms of this cultivar. Surrounded by dark green leaves, the appearance of lightness and chroma in the flowers is enhanced through simultaneous contrast.
A second photograph, by Dr. Herbert Spady (Plate 219), portrays 'Harvest Moon', one of the parents of 'Creole Belle'. A connection between the deep red color near the corolla base in 'Creole Belle' and the greenish yellow throat of 'Harvest Moon' is admittedly speculative. The appearance of green in plant tissues is associated with the presence of chlorophyll in the chloroplasts of the cells. The presence of green coloration is usually most evident near the base of a corolla, the area of attachment to the receptacle. In 'Creole Belle', the presence of chlorophyll in this area would contribute to the deep red color. If there is a substantial presence of chlorophyll, light rays with yellowish green wavelengths reflected from the chloroplasts would combine in the visual system with rays carrying the blue and red wavelengths reflected from the purplish red pigmentation in cell vacuoles. The integrated effect would be a darkening of the perceived color and a shift in hue toward red.
The hypotheses suggested by the photographs from Rhododendron Hybrids need testing. It would be useful to examine petals from 'Glowlight' as well as from 'Creole Belle' to determine whether the epidermis is coated with cutin and whether these cultivars differ significantly from others in this respect. Further use of microscopy should reveal whether chloroplasts are plentiful in cells near the base of the corolla.
The best of efforts are at times frustrated by the vagaries of nature. For one year, I thought that I had solved the problem of what causes the brilliance of 'Creole Belle'. But in the spring of 2000, when I examined the blossoms of my plant, no white cells were seen at any time during the blooming period. I traveled 60 miles to the south to a friend's garden and found that the flowers on his 'Creole Belle' had the same white cells, in the same order of layers, that my plant has in 1999. His plant was in full sun; mine was in shade. The spring of 2000 in my area was overcast and daytime temperatures never rose above 55° F. for months; nights were in the 30s and 40s. This is a good example of the genetic/cultural interactions described by Griesbach (4).
This experience confirmed my conclusion that the white color in the cells of 'Creole Belle' is not caused by solid bodies. It also shows the extent to which the vagaries of environment - for example, temperature, sunlight, and availability of nutrients - can in little - understood ways affect flower coloration. The anthocyanins and flavonoid co-pigments are water soluble and bond with sugars in the cell vacuole. The expression of color depends in part on environmental conditions that affect the availability of the sugars and the degree to which these combine with the flavonoids. Clearly, a number of intriguing issues await research. Those of greatest interest to me relate to the processes of human perception of the physical variables existing in these plants.
2
'Creole Belle' Plant Name Register
'Creole Belle': Elepidote. ('Vulcan' x 'Harvest Moon'). Hybridized (1968), raised, named, introduced (c 1979, through Willard Thompson Nursery), and registered (1990) by Willard and Margaret Thompson, Waldport, OR. Described by W & M Thompson with D W Goheen, Camas, WA. Widely funnel-shaped flowers are 5" to 5½" across x c 3" long, with 6-7 wavy-edged lobes. The corolla is strong purplish red (64C) with a deep red (53A) flare in the center, and deep red (53A) spots on the dorsal lobe. The cardinal red calyx is less than ⅛" long. The dome-shaped truss of 16-18 flowers is 8" wide x 5" to 6" high. Leaves, which are held 3-4 years, are elliptic, flat, acute at apex, cuneate at base, 6" to 7" long x 1½" to 2" wide, dull green, and hairless. The plant is very floriferous, blooms in early May, is 8' high x 8' wide in 15 years from seed, and is upright, with a dense growth habit. Plant and buds are hardy to at least –5°F and 0°F, respectively.
Acknowledgements
Dr. Anu Singh Cundy of the Biology Department at Western Washington University helped me by giving me advice, the use of her laboratory and equipment, instruction in her classes, evaluation of my slides, guidance in readings in books and journals and encouragement. She was sine qua non.
My technical reviewers were Donald Voss and Dr. Robert J. Griesbach, both of whom helped me in many ways. They enabled me to avoid blind alleys, hastened my progress, gave good suggestions and identified my errors. I am much indebted to them.
Dr. Carl L. Withner, author of six books on orchids, not only encouraged me but also aided me in interpreting my slides. The method of slide preparation I used generated many confusing slides difficult to interpret.
References
1. Arnheim, R. 1974.
Art and Visual Perception
. Berkeley, CA: University of California Press, New Version 1974, pp. 305-320, 477.
2. Berns, Roy S. 2000.
Billmeyer & Saltzman's Principles of Color Technology, Third Edition
. New York: John Wiley & Sons.
3. Capon, Brian. 1990.
Botany for Gardeners
. Portland, OR: Timber Press.
4. Griesbach, R. 1987. Rhododendron flower colors: genetic/cultural interactions,
Journ. Amer. Rhod. Soc.
, Vol. 41, No. 1, pp. 20-21.
5. Hardgrove, D. L. 1966. Hybridizing experiences and recommendations,
Journ. Amer. Rhod. Soc.
, Vol. 20, No. 4, p. 216.
6. Itten, J. 1970.
The Elements of Color
, New York: Van Nostrand Reinhold, pp. 32-63.
7. Judd, Deane B., and G. Wyszecki. 1975.
Color in Business, Science, and Industry
, New York: John Wiley & Sons, pp. 5-34, 398-399, 408, 372.
8. Kaiser, P. K., and R. M. Boynton. 1996.
Human Color Vision, Second Edition
, Optical Society of America, pp. 92-120.
9. Katz, David. 1935.
The World of Color
. Reprinted 1970. London: Kegan, Paul, Trench, Trubner, pp. 13, 160, 264.
10. Kirk, John T.O., and R.A. Tilney- Bassett. 1978.
The Plastids, Revised Second Edition.
Amsterdam: North-Holland Biomedical Press.
11. Mol, J., E. Grotenwald and R. Koes. How genes paint flowers and seeds,
Trends in Plant Science
, Vol. 3, No. 6, pp. 213-214.
12. Murray, J. 1990. American Rhododendron Society Plant Registry,
Journ. Amer. Rhod. Soc.
, Vol. 44, No. 4, p. 234.
13. Murray, J. 1991. American Rhododendron Society Plant Registry,
Journ. Amer. Rhod. Soc.
(insert) Vol. 45, No. 2, p. 6.
14. Noda, K., B. Glover, P. Linstead, and C. Martin. 1994. Flower colour intensity depends on specialized cell shape controlled by a Myb related transcription factor,
Nature
, Vol. 369, No. 23, pp.661-664.
15. Percherer, B. 1992. The color of rhododendron flowers,
Journ. Amer. Rhod. Soc., Vol
. 46, pp. 200-202.
16. Raven, R.H., R. F. Evert and S. E. Eichhorn. 1992.
Biology of Plants, Sixth Edition
, New York: W. H. Freeman Co., pp. 636, 906.
17. Royal Horticultural Colour Chart, London, Royal Horticultural Society, 1995 edition.
18. Salley, Homer E., and Harold E. Greer. 1992.
Rhododendron Hybrids, Second Edition
, Portland, OR: Timber Press.
19. Spethman, W. 1980. Flavonoids & cartenoids of rhododendron flowers and their significance for the classification of the genus
Rhododendron
. In
Contributions Toward a Classification of Rhododendron
, ed. J. L. Luteyn, Bronx, NY: The New York Botanical Garden, p. 338.
20. Swain, T. 1976. Nature and Properties of Flavonoids. In
Chemistry and Biochemistry of Plant Pigments, Second Edition
, ed. T.W. Goodwin, New York: Academic Press, Vol. 1, p. 426, Vol. 2, p. 459.
* Name is not registered
Notes
1
Apparently 'Glowlight' was never registered. If any ARS member has a plant of 'Glowlight', please contact.
2
Since this manuscript was reviewed by my peer reviewers, I have discovered several references about the role of cell shape in creating flower colors. Researchers (11) have found, in addition to vacuolar pH and co-pigmentation, the shapes of petal cells affect the color perceived. Other researchers (14) have discovered that flower color intensity depends on specialized cell shape. My own belief is that the concavity of the interior surfaces of cells and their wetness because of cell sap must also be involved. This may, however, be part of what they mean by cell shape.
Dr. Ching, a member of the Komo Kulshan Chapter, is a psychologist with a special interest in the interrelations of science and art.