The Rhododendron Leaf Scale
Clifford Desch, Jr., West Hartford, CT
To most non-rhododendron oriented gardeners the term "scale" conjures up an image of an insect plant pest. To the rhododendron grower, a "scale" is an anatomical feature characterizing the lepidote group of this genus. However, few of these growers have observed leaf scales in histological sections or are familiar with their microscopic structure. In a monograph on the Rhododendron leaf trichomes (hairs and scales), John Cowan (1950) reports detailed descriptions of the epidermal scales. Unfortunately, this text is out-of-print and, therefore, unavailable to most members of the American Rhododendron Society. I decided to investigate these structures first hand and report my observations on three species below.*
Pieces of leaves of
Rhododendron carolinianum
(Subsection Caroliniana),
R. edgeworthii
(Subsection Edgeworthia) and
R. oulotrichum
(Subsection Trichoclada) were fixed in Craf #5 solution, embedded in paraffin-piccolyte, sectioned at 10 µm
1
and stained with Safranin 0 and Fast Green. Additional
R. carolinianum
leaf tissue was fixed in glutaraldehyde, post-fixed in osmium tetroxide, embedded in Spurr's low viscosity epoxy resin, sectioned at 1-2 µm and stained with Methylene Blue-Azure II.
Scales are multicellular structures of epidermal origin consisting of a stalk and an expanded cap (Fig. 1). The cuboidal cells of the stalk are small and contain living cytoplasm. They are compressed in the distal portion of the stalk. Cap cells are large, variable in shape and do not contain living cytoplasm. The scales are set in depressions in the leaf with their caps extended slightly beyond the leaf surface. The caps cover the depression and, thereby, also cover a ring of stomatal apparati which are positioned around the scale stalk in the depression (Fig. 2). This arrangement, presumably, reduces the air velocity over the stomata, however, stomatal apparati are not restricted to the leaf scale pits.
Five scale types are recognized by Cowan (1950): entire, lacerate, crenulate, undulate and vesicular. These designations are based on the outline of the scales when viewed from above and the shape of the outermost row of cells around the cap (rim or ray cells). The "entire" scale is the most common type being found in all but 6 "series" of the lepidote group (excluding Vireya).
|
|
|
|
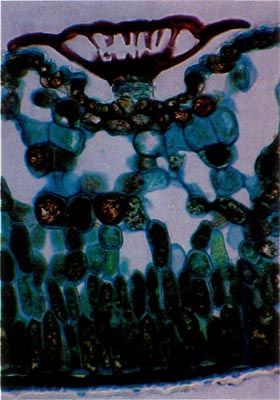
Fig. 1.
R. carolinianum
.
Paraffin; cross-section through leaf. Scale sits in a depression on the lower leaf surface. Note absence of cytoplasm in cap and a stomatal apparatus to each side of the stalk. |
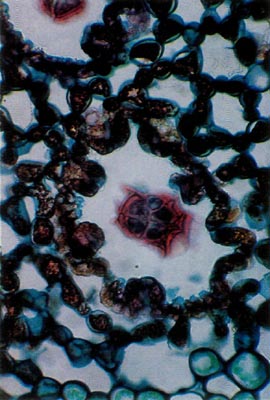
Fig. 2.
R. carolinianum
. Paraffin;
section parallel to leaf surface. Scale stalk in center of pit surrounded by 8 stomatal apparati. |
|
| photos by Clifford Desch, Jr. | ||
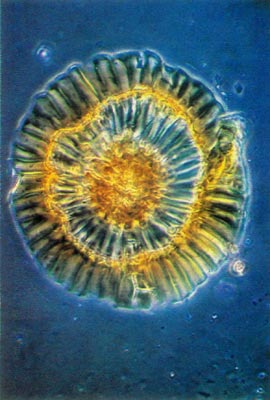
Rhododendron carolinianum possesses scales of the entire type (Figs. 1 and 2). The stalk cells are stacked in 4 or 5 layers with 4 cells per tier. The cap consists of 4 central, columnar cells positioned directly over the stalk. These are surrounded by a ring of 10-15 columnar cells and they are surrounded, in turn, by another concentric row of 20-25 cells, the rim cells. These latter cells are the largest with the cell wall of their peripheral edge expanded radially 30-40 µm. The combined effect of the extended edges of adjacent rim cells creates a thin, flattened rim around the cap. Prior to death of the rim cells, the cytoplasm resided in the region of the cell nearest the center of the scale cap. When viewed from above, the scales are nearly circular ranging from 150-250 µm in diameter (Fig. 3).
|
|
Fig. 3.
R. carolinianum
.
Whole mount of scale. Note rim and compare with Fig. 1. photo by Clifford Desch, Jr. |
Scales also occur on the upper leaf surface but are outnumbered approximately 20 to 1 by those on the lower surface. The upper scales differ from the lower ones only by having thinner cell walls and they reside in shallower pits which lack stomata. In fact, stomata are absent on the upper leaf surface.
The scales of
Rhododendron edgeworthii
are also of the entire type (Fig. 4). They are similar to those of
R. carolinianum
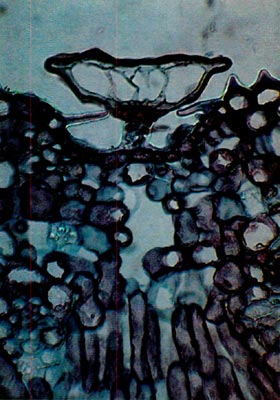
except, being of a small cap size, they do not completely occlude the opening of the depression in which the scale sits. Also, the rim cells of the cap do not possess the marked cell wall extension. They range in cap diameter from 110-140µm.
|
|
Fig. 4.
R. edgeworthii.
Paraffin;
cross-section through leaf. Scale does not completely occlude the pit. One stomatal apparatus is visible to the left of the scale. photo by Clifford Desch, Jr. |
Rhododendron oulotrichum displays vesicular scales. These are mushroom or bladder-shaped, lack elongate peripheral cap cells and are circular when viewed from above. (Figs. 5 and 6). The caps vary in diameter from 80-210 µm. In addition to the Trichocladum Series, vesicular scales are found in the Campylogynum, Boothii and Glaucum Series. The latter two series also possess entire type scales.
|
|
|
|
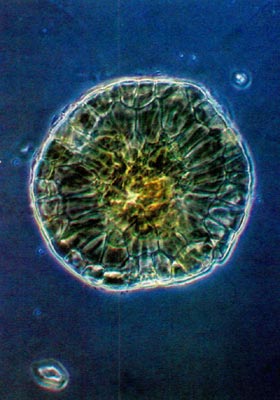
Fig. 5.
R. oulotrichum
.
Whole mount of scale. Compare rim to that in Fig. 3. |
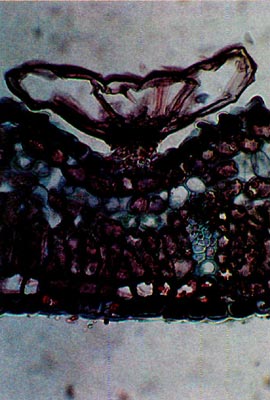
Fig. 6.
R. oulotrichum
. Paraffin;
cross-section through leaf. |
|
| photos by Clifford Desch, Jr. | ||
My brief study did not include observations of lacerate, crenulate and undulate scales. Cowan (1950) reports the lacerate scales of the Anthopogon and Cephalanthum Series possess a cap margin which is irregular and deeply indented (viewed from above) because the two radial walls of each rim cell are of unequal length.
Crenulate scales are found in the Saluenense and Lepidotum Series with the latter also having entire scales. Each rim cell of the crenulate type is rounded at its "peripheral" end thus, giving the margin of the scale cap a scalloped effect.
Undulate scales appear in the Lapponicum Series (plus entire scales) and in the Vaccinoides Series. The scale cap margin is undulate due to the irregular length of the rim cells.
As an animal taxonomist specializing in parasitic hair follicle mites of the Family Demodicidae, I am well aware that new information and in-depth study of a taxonomic group, be it animal or plant, often necessitates rethinking and realigning intrageneric relationships. The large, diverse, widespread genus
Rhododendron
is no exception. In newer versions of
Rhododendron
classification, detailed studies of such microscopic features as the above described scales of the plants are being utilized to aid in the redefinition of the "series" into more natural and rational Subsections (see Cullen and Chamberlain, 1978).
Note: Since submitting this paper in May 1980, Dr. J. Cullen of the Edinburgh Royal Botanic Garden has revised the taxonomy of the lepidote Rhododendrons (Cullen, 1980). Cullen concurs with Duncan and Pullen (1962) that
Rhododendron carolinianum
and
R. chapmanii
are synonymous with
R. minus
Michaux 1772. The specific epithet
minus
has priority according to the rules of nomenclature. These authors, however, recognize two varieties: var.
minus
(=
R. minus
and
R. carolinianum
) and var.
chapmanii
(=
R. chapmanii
). More recently, Hardin and Gensel (1982) studied the leaf scales of
R. carolinianum
,
R. minus
and
R. chapmanii
with the aid of the scanning electron microscope. They could not distinguish interspecific morphological differences and, thus, feel their findings add support to the concept of a
R. minus-complex.
References:
Cowan, J.M. 1950. The Rhododendron Leaf. Oliver and Boyd, Edinburgh and London, pp. 120.
Cullen, J. 1980. A revision of
Rhododendron
I. Subgenus Rhododendron sections Rhododendron and Pogonanthum. Notes. R. Bot. Gard. Edinb. 39: 1-207.
Cullen, J. and D.F. Chamberlain. 1978. A preliminary synopsis of the genus
Rhododendron
. Notes R. Bot. Gard. Edinb. 36: 105-127.
Duncan, W.H. and T.M. Pullen. 1962. Lepidote rhododendrons of the southeastern United States. Brittonia 14: 290-298.
Hardin, J.W. and W.H. Gensel. 1982. Atlas of folar surface features in woody plants, IV. Rhododendrons (Ericaceae) of eastern North America. Bull. Torrey Bot. Club. 109: 189-198.
1 1 µm = 1/1000 mm = 1/25,400 inch
Note: Dr. Hedegaard presented a paper on his work the Morphological Studies of Rhododendron Fruit, Seeds and Seedlings at the International Rhododendron Conference in 1978. This paper was published in 1980 "Studies in the Genus Rhododendron, Johannes Hedegaard. G.E.C. Gads Publishing House.