Polyploids in Rhododendron Breeding
August E. Kehr
Hendersonville, North Carolina
I have been interested in polyploidy ever since I first learned of the subject as a graduate student in the 1940s. While this fascinating subject has not been a central part of my professional life's work, it has been an abiding interest in retirement.
In this paper I will discuss the basics of polyploidy, development of plants new to science by polyploidy, recent examples of polyploidy in rhododendrons and azaleas, improved methods of producing polyploids and examples of recent hybridization using polyploid plants.
Synopsis
Polyploid plants are a source of most of the world's food supply. They are fundamental in the improvement of crop and ornamental plants. Polyploid forms of rhododendrons and azaleas, reported since 1953, are described and references cited. An improved technique for producing polyploid plants by colchicine treatment of seedlings is described. It is shown how such new polyploids are capable of extending opportunities for rhododendron and azalea improvement.
All living things are made up of cells. Within the cells are distinct entities called chromosomes. Commonly there is one set of chromosomes from each parent, or two sets of chromosomes in a cell. Such organisms therefore have a two-fold number of chromosomes, and they are referred to as diploid ("di" meaning two). Any plant with more than two sets of chromosomes in its cells is called a polyploid ("poly" meaning many). Thus a polyploid is any plant with more than two sets of chromosomes in its cells.
Polyploidy has many beneficial effects on plants. From a horticultural viewpoint one of the greatest values is that they have much larger and heavier textured flowers. Because of their heavier texture, the flowers last longer than the normal flowers. Lesser value is that the leaves are larger and the twigs are thicker. Polyploids also have a complex Mendelian genetic ratio, making it nearly impossible to project the characteristics to be expected in their progenies. Likewise polyploids most commonly flower later in the season than related diploids. In addition, the plants grown from seed usually require a longer time to flower for the first time.
Whether we realize it or not, polyploids provide most of our daily food. Of the world's 10 most important food crops
1
, most are polyploids. Almost all of our vegetables and fruits are of polyploid origin. In many of these crops scientists have conducted experiments to reconstruct the historic formation. For example, wheat arose from the combination of three natural wild species. This has been demonstrated by artificially repeating that process (1). Today's sweet potatoes also arose from the gradual combination of the total chromosomes of three wild species (2). Many more of our commercial plants resulted from the increase in chromosome number by the process of polyploidization.
Another example of polyploidy can be seen in experiments with
Nicotiana
. There are about 60 species of
Nicotiana
, one of which is
Nicotiana tabacum
, the ordinary plant used for smoking tobacco. At one point I developed a Nicotiana plant with a huge number of chromosomes to see what would happen when a single plant was encumbered with an excessive number of chromosomes and whether or not it would collapse from sheer numbers of chromosomes. The plant had 144 chromosomes and an unusual appearance but was fully fertile despite all of its excess chromosomes. This experiment was carried further by Dr. H.H. Smith of Brookhaven National Laboratory, who selfed the plant for 10 generations (3). Because the plant had so many excess chromosomes beyond its vital needs, the superfluous ones were lost as the selfing process continued. The final result of the experiment was the formation of three distinctive races, each having 108 chromosomes. All three races differed in their genetic behavior from each other and also from all of their original parents. If found in the wild, these would have been considered new species.
Recent Examples of Polyploidy in Rhododendrons and Azaleas
In 1953 three Japanese scientists reported that four evergreen azaleas, commonly grown in Japan, were polyploids (4). These were 'Bangaku' (39 chromosomes) and 'Banka', 'Taihei' and 'Wako'(52 chromosomes). The Japanese cultivar 'Getsutoku' is probably also a tetraploid with 52 chromosomes.
In 1956 a comprehensive report on the chromosome numbers of many species of rhododendrons, as researched by Dr. Janaki Ammal (an Indian student in England), was reported (5). Unfortunately, very little of this monumental work by Dr. Ammal is found in the literature in any detail.
In 1971 I reported the development of a tetraploid form of
Rhododendron carolinianum
(now
R. minus
var.
minus
Carolinianum Group) and registered it under the name of 'Epoch' (6). To my knowledge this was the first tetraploid form of
Rhododendron
developed as the result of treatment with colchicine
2
reported in the literature.
In 1972 seven tetraploid forms of seven different azalea cultivars were released by scientists at USDA at Beltsville, Md. The seven were made polyploid by treatment with colchicine. None of this information was preserved in the literature. The seven were designated by [unregistered] names as follows: 'Anytime Tetra', 'Casablanca Tetra', 'Chimes Tetra', 'Guy Yerkes Tetra', 'Hershey Red Tetra', 'Pink Gloria Tetra' and 'Road Runner Tetra' (probably lost entirely).
These seven tetraploids were released to selected nurserymen for evaluation and increase. I have three in my garden but don't know the location of any others in this country. I had sent material to Mr. Jack F. Wilson, Victoria, Australia, who still has the plants. Through his kindness most of these valuable plants have been re-introduced into this country.
Other tetraploids that are apparently not reported in the literature include a tetraploid form of
R. fortunei
by Dr. Max Byrkit, Williamsport, Md., and a tetraploid form of the hybrid of ('Idealist' x 'Crest') named 'Queen of McKenzie' by Dr. Carl H. Phetteplace. This latter plant came to me through the courtesy of Jack Rosenthal. The Halfdan Lem hybrid 'Jingle Bells' may also be a tetraploid because it breeds like one and also has flowers with extremely heavy substance.
I have developed a tetraploid evergreen azalea (#315-1) that is cream-colored from the selfed cross of ('Perle de Swynaerde' x Pryor 75-305). This plant carries the genes for flavanoid pigments and has been the central focus of my most recent work in developing a yellow-flowered evergreen azalea. 'Perle de Swynaerde' is the only evergreen azalea I know that is recessive for white color. When this cultivar is crossed with any color, the progeny color is not influenced by hidden color genes.

|
|
Figure 1.
Two dividing cells in a plant, at a stage when accurate
chromosome counts can be made. This is from a daylily, and one can count the chromosomes in the dividing cell to the left. |
Improved Methods of Production of Polyploids
The most recent advance in polyploidy is being done by Steve McCullock of the Briggs Nursery in Olympia, Wash. He has perfected the technique of treating tissue culture with colchicine to develop polyploids. He has succeeded in developing a sizable number of polyploid forms of rhododendrons and azaleas. This work marks one of the greatest advances in the improvement of these plants in decades.
Beginning about 1984, I developed some improvements in the conventional methods of using colchicine to treat seed of magnolias. These improvements permitted me to produce over 50 polyploid magnolia trees and more recently rhododendrons and azaleas (7), (8)
The primary action of colchicine is that it apparently prevents the formation of a cell wall in dividing cells. Thus the cells divide their chromosomes in a normal manner, but the colchicine prevents the formation of the immediate subsequent dividing cell wall, and hence all the just-divided chromosomes remain in the same cell. In the next division of that cell, all the chromosomes likewise remain in the same cell, thereby resulting in increasing the number of chromosomes by a factor of two. The dominant cell in the growing point
must be polyploid
if the polyploid condition is to persist in subsequent growth of the plant.
Although colchicine treatment of woody plants has been described for over 50 years, relatively few woody plants have been reported to date. Most of the older methods consisted of first germinating the seeds on filter paper or blotting paper. After germination was underway, the seedlings were treated with an aqueous solution of colchicine. Such treatments therefore affected
all
parts of the germinating seedlings, the shoots as well as the roots. Such treatments affected the roots so adversely that in most cases the seedlings did not survive.
The improved method is only a slight change, but it makes a world of difference in the final results. It starts with making a stock solution of 1 gram of colchicine in 100 cc of water (about 6 oz.). This 1% stock solution lasts for several years, depending on its use. The working solution is made of 20 cc of stock solution and 180 cc of water (to make a 0.1 % concentration) to which 1 cc of dimethyl sulfoxide
3
(commonly called DMSO), and 1/10 drop of dish washing fluid is added. The 1/10 drop is made by diluting one drop of dish washing fluid in 10 drops of water and then using one drop of the diluted dish washing solution. Too high a concentration of the dish washing fluid is injurious to the seedlings. The DMSO increases the permeability of the cell walls and thereby increases the entrance of the colchicine into the cells of the growing point, while the dish washing fluid "wets" the surface of the cells. These two chemicals jointly increase the effectiveness of the solution.
Seeds are planted in the normal manner on the substrate of choice. When the cotyledons of the germinating seed are well developed, and just before the true leaves are visible, the above mixture of colchicine, DMSO and dish washing fluid is fine misted on the developing seedlings until a minute amount collects between the two cotyledons. The seedlings are misted twice daily for at least 7-10 days and until the true leaves are plainly visible. The seedlings should have at least an 18-hour day and be kept in a chamber that maintains as near 100% relative humidity as possible. By this method the roots are not affected, and the seedlings grow normally. The seedlings that have become polyploid will have a lower length-width ratio than the diploid seedlings. In brief, the leaves are wider and thicker than the undoubled seedlings. For very small seeds, such as rhododendrons and azaleas, these differences are more noticeable after the first transplanting.
This method (as described) will work on all dicotyledonous plants but not on monocotyledonous plants.
Colchicine is available from most scientific houses, but a business letterhead may be needed to purchase it. One company that sells it is Sigma Chemical Company, P.O. Box 14508, St. Louis, MO 63178-9916. Your local college or high school chemistry department could probably help you in finding sources.
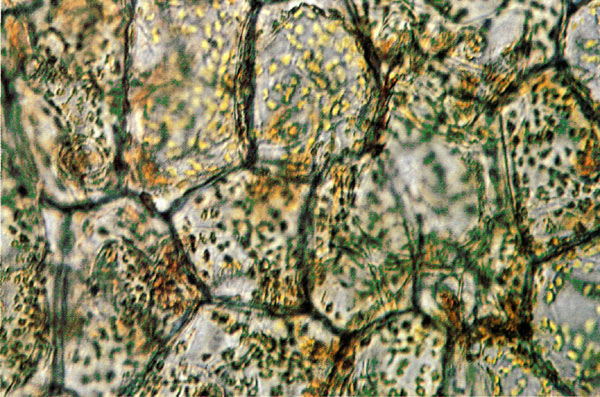
|
|
Figure 2.
Photomicrograph of carotenoid plastids in
a flower of a yellow colored R. calendulaceum . |
Examples of Recent Hybridization Using Polyploid Parents
Many crosses can be made with polyploid parents that are not successful at the original diploid level. For example, it is virtually impossible to hybridize
R. minus
var.
minus
Carolinianum Group with
R. augustinii
using the diploid form of the former. If one succeeds in making this cross, the offspring will be fully sterile. However, if one makes the cross of
R. augustinii
(seed parent) by tetraploid
R. minus
var.
minus
Carolinianum Group 'Epoch' (pollen parent), one gets lots of seed and the seedlings will all be fertile. When diploid forms of evergreen azaleas are likewise used as pollen parents, such crosses are difficult or impossible to make.
Some examples of unusual crosses made in 1995 are as follows:
(a) Crosses using pollen of tetraploid
R. fortunei
: (
R. calendulaceum
x
R. fortunei
'Tetra'*).
(b) Crosses using pollen of evergreen azalea #314-1: (
R. augustinii
#891 x evergreen azalea #314-1) (a promising cross.); (
R. calendulaceum
x evergreen azalea #314-1).
4
References
1. Darlington, CD. In chromosome botany. Hafner Press, p. 162; 1973.
2. Ting, Y.C.; Kehr, A.E.; Miller, J.C. A cytological study of sweet potato plant,
Ipomea batatas
(L) Lam and its related species. Am. Naturalist 91 (858): 197-203; 1956.
3. Smith, H.H.; Stevenson, H.O.; Kehr, A.E. Limits and consequences of multiple alloploidy in
Nicotiana
. The Nucleus 1(2): 205-222; 1958.
4. Hosoda, T.; Moriya, A.; Sarashima, S. Chromosome number of Satsuki,
Rhododendron lateritium
. Genetica 26: 407-409; 1953.
5. Darlington, CD.; Wylie, W.P. Chromosome atlas. New York: Macmillan Co. pp. 217-218; 1956.
6. Kehr, A.E. A tetraploid
Rhododendron carolinianum
. Am. Soc. Quart. Bull. 25 (1): 4-7; 1971.
7. Kehr, A.E. Magnolia improvement by polyploidy. Magnolia 26 (2): 26-28; 1991.
8. Kehr, A.E. Polyploidy in woody plants. Am. Nurseryman 183 (3): 40-47; 1996.
1
Corn, wheat, millet, oats, yams, sweet potatoes, soybeans, barley, potatoes, rice.
2
Colchicine is a dangerous chemical and must be handled with great care. Do not allow it to get on your skin or in your eyes. Wash it away immediately if it does. Follow label precautions carefully.
3
DMSO: Do not breathe fumes or vapors, do not ingest. Follow label precautions carefully.
4
Actual chromosome counts or chemical methods were not used to determine polyploidy in the polyploid plants mentioned in this article. Polyploidy was determined by visual means only, fully recognizing that such morphological assessments are not always accurate beyond a reasonable doubt. The length-width ratio of the leaves, leaf texture, leaf appearance, and subsequent breeding behavior are means of assessment of the polyploid state that are available to every rhododendron grower to whom the more critical and more accurate methods are not available.
* Name is unregistered.
Dr. Kehr, a member of the Southeastern Chapter, authored the article "The Mysterious Red Maximum from Mt. Mitchell" in the winter 1994 issue of the Journal.