Why Do Some Hymenanthes Rhododendrons in Toronto Raise and Lower their Leaves Seasonally?
Willem Morsink
Toronto, Ontario
Canada
Synopsis
Hardy rhododendrons in Toronto move their leaves downward in autumn apparently to prevent high light exposure damage during freezing episodes. Tender cultivars position their upper leaves downward only to a more or less horizontal plane, possibly to compensate for decreasing light intensity while continuing photosynthesis. Seasonal leaf angle changes through a calendar year can be taken to obtain a preliminary indication of cold hardiness or lack thereof for new cultivars, as illustrated in Table IV.
Introduction
Previously, the author surveyed the hardiness of rhododendrons in Ontario from Niagara Falls, Ontario, to Montreal Quebec (4), to find out what Ontario's gardeners had cultivated during the fifty years preceding 1995. However, many hybrids from the Atlantic region and Finish and other hybrids were not part of the survey. As an alternative to field testing these hybrids, the numerous eco-physiological papers on
Rhododendron
by Professor Erik Nilsen, (1, 2a, 2b, 3, 5, 6, 7, 8, 9, 10, 11, 12, and 13), and papers by many others as reviewed by Vainola (15), provide research methods and approaches that may be used to establish preliminary cold hardiness evaluations for new hybrids. Of special interest are the two physiological, distinct winter-leaf-movements in
Rhododendron
, i.e., leaf freeze-curling and change in leaf angle (5), and the fact that intensity of these two movements is associated with cold tolerance (10, 11, and 13).
The large-leaf evergreen rhododendrons in genus
Rhododendron
, subgenus Hymenanthes, which are shade-adaptive woody plants of temperate mountainous regions, are sensitive to excessive sunlight that may damage the photosynthetic pigments (2a). Inhibition of photosynthesis (photo-inhibition) can occur when excessive amounts of light reach the leaves in exposed high-light (high-irradiation) environments during the growing season. Photoinhibition may also occur during sunny freezing periods in the dormant season when the sunbeam heats the leaves (sun flecks), which causes rapid changes in leaf temperature and freeze-thaw damage (1, and 11).
Nilsen (12) concluded that changes in leaf angle protect against photoinhibition, and leaf curling protects against freezing damage by rapid thaw.
|
Table I. Intense freeze-leaf-curling from 0 to 100 percent (N1), for selected rhododendrons in Toronto (N2), Canada.
Notes: N1 = Percent leaf curling = [1-(residual freeze leaf width/: leaf width at +1 C air temperature) x 100]. N2 = Toronto locations; #1 = author's garden and #2 = Edwards Gardens. |
|||||
| Air Temperatures Celsius | |||||
| Leaf width | +1 | -1 | -9 | -15 | |
| Rhododendron | cm | Leaf curl percentages | |||
| #1 PJM Group | 2.3 | 0 | 11 | 78 | 78 |
| #1 'Calsap' | 4.6 | 0 | 14 | 78 | 83 |
| #2 'Swansdown' | 4.3 | 0 | 6 | 79 | 82 |
| #1 'Haaga' | 3.8 | 0 | 48 | 78 | 80 |
| #1 'Helsinki University' | 4.5 | 0 | 52 | 77 | 80 |
| #1 'Saint Michel' | 3.9 | 0 | 45 | 71 | 80 |
| #2 'Cadis' | 5.0 | 0 | 5 | 75 | 80 |
| #2 'Janet Blair' | 4.8 | 0 | 11 | 63 | 79 |
| #2 'Roseum Elegans' | 4.3 | 0 | 8 | 74 | 76 |
| #2 Yak hybrid | 3.0 | 0 | 8 | 75 | 75 |
| #1 'Catawbiense Album' | 4.2 | 0 | 8 | 67 | 75 |
| #2 'Lodestar' | 6.6 | 0 | 15 | 73 | 74 |
| #2 'Dexter's Champagne' | 4.3 | 0 | 12 | 71 | 74 |
| #1 'Nova Zembla' | 3.7 | 0 | 3 | 62 | 66 |
| #2 'Nova Zembla' | 3.9 | 0 | 3 | 65 | 71 |
| #2 'Catawbiense Boursault' | 5.1 | 0 | 0 | 65 | 70 |
| #2 'Anah Kruschke' | 5.0 | 0 | 5 | 60 | 60 |
| #1 R. brachycarpum as ssp. tigerstedtii | 2.9 | 0 | 17 | 65 | 64 |
| #2 R. metternichii (now R. degronianum ) | 2.3 | 0 | 11 | 67 | 67 |
| #1 R. smirnowii | 3.8 | 0 | 7 | 60 | 70 |
| #2 R. makinoi | 2.5 | 0 | 20 | 65 | 65 |
| #1 R. maximum 'Mount Mitchell' | 4.3 | 0 | 6 | 56 | 71 |
| #1 R. maximum (Tunnel Mt., VA) | 4.1 | 0 | 12 | 59 | 66 |
| #1 R. sutchuenense | 4.1 | 0 | 26 | 80 | 82 |
| #2 'Golden Genie' | 3.7 | 0 | 0 | 76 | 79 |
| #1 'Sumatra' | 4.4 | 0 | 5 | 65 | 67 |
| #1 'Cunningham's White' | 3.4 | 0 | 0 | 69 | 70 |
Freeze-Leaf Curling
Nilsen noted that winter leaf curling is mainly stimulated by freezing temperatures, and only partly by low leaf water potential: "Water potential is determined by soil temperature, soil water availability, and water uptake by roots, hydraulic conductance through stems, and leaf vapor pressure, among other factors." Summer leaf curling, in contrast, is induced by water stress, but the curling may be only 30- 45% as compared to freeze-leaf curl in the winter (7). Winter freeze curling possibly reduces the rate of re-warming following freezing of leaves (10). The physiological and anatomical factors that regulate leaf curling are complex and are not yet well understood (12).br> Cold hardy rhododendrons with intense cold induced leaf curling lose cell water and cell turgor pressure easily, starting at small water deficits and leaf temperatures ranging from 0C to 2C. Conversely, cold sensitive species that showed only slight curling, or no leaf curling and leaf angle movement down to 20C, retained more cell water, and more turgor cell pressure under water stress conditions during the dormant season (13).
Rapid freeze-curling species were noted as
Rhododendron catawbiense
,
R. maximum
,
R. minus
,
R. micranthum
, (13) and
R. brachycarpum
(15). Although cold hardy hybrids curled strongly and cold sensitive hybrids showed little leaf or no curling (11), cold hardiness did not consistently relate to winter-freeze-leaf-curling, when Nilsen (13) evaluated seventy-five hybrids.
Freeze-leaf-curling and freeze-leaf-angle changes, in Toronto, occur together for the more cold hardy rhododendrons. These movements occur during winter freezing periods during the day as well at night. Freeze-leaf-curling information, shown in Table I and Plate I-1,-2,-3 and -4, has therefore been included for several rhododendrons, some of which were also used in the observations on leaf angle changes in Toronto.

|
|
Plate I-1, -2, -3, and -4. Freeze-curled and vertically down-turned leaves during mid-winter freezing episodes at -10C.
Elepidotes: 1 = R. brachycarpum as ssp. tigerstedtii seedling; 2 = R. 'Calsap'. Lepidotes: 3 = PJM Group; 4 = R. 'Windbeam' shows only freeze curled leaves. |
Dormant Season Leaf Angle Changes
Nilsen and others (1, 9 and 12) noted that changing the leaf from a horizontal to a vertically downward position may serve to protect the photosynthetic apparatus of rhododendron leaves from high light damage during freezing periods. Especially during sunny winter freezing episodes, leaf damage can become excessive and mortality may result.
Species with long petioles showed stronger leaf angle changes than species with short petioles (11). Since petioles are not necessarily in equilibrium with the water deficit in the leaf blade and the petioles have thin cuticles, they dehydrate easily; as a result the leaves become pendant with loss of turgor pressure (7). When the temperature rises above freezing the leaf blade (lamina) first uncurls and then rises to a more horizontal position. Nilsen (11) concluded that the intensity of leaf angle change correlates with cold tolerance.
In addition to winter leaf movements, one can observe that many of the Toronto-hardy rhododendrons also change leaf angle substantially before and after the winter dormant season. This begged the question: "Why do many of the large-leaf-evergreen Hymenanthes rhododendrons in the Toronto region raise and lower their leaves through the four seasons as expressed through a change in leaf angles?" This question prompted the leaf-angle-change explorations from June 21, 2002, to June 21, 2003, and again for a second year to June 21, 2004, in the author's Toronto garden.
Location of the Toronto Garden
The measurements of seasonal changes in leaf angles were made in the author's small rhododendron garden. This garden is situated just north of Toronto's major east to west highway, #401. Leaf freeze-curl measurements were taken in this garden and in the Edwards Gardens, which is located just south of the #401.
This highway provides an approximate H1-H2,
Rhododendron
hardiness divide (4), for the Toronto region with the milder Lake Ontario H2 hardiness area to the south and the much colder H1 area in the winter to the north of the #401 highway.
|
H = LOCATION COLDNESS (HARDINESS)
(from reference 4)
* The January temperature is the normal minimum for the coldest month, not the extreme low temperature used in the USA. ** Mdd's are a "measure of coldness" for the duration of a dormant season. Mdd's are expressed as "minimum dormant degree days" below 41F = 5C, for locations. |
|||
| January* "H" | Mdd's** | ARS H | USDA ZONES |
| H1 = to -16C (+4F) | 2300 (C), or 4500 (F) | H1 = -25F(-32C) | 4b/5a |
| H2 = to -11C (+13F) | 1800 (C), or 3500 (F) | H2 = -15F(-26C) | 5b/6a |
| H3 = to -07C (+20F) | 1300 (C), or 2500 (F) | H3 = -05F (-20C) | 6b/7a |
Canopy situation
Shading was found to change the irradiance and spectral composition of light, and shading increased the longevity of leaves of
Rhododendron maximum
in natural habitats with mainly overhead deciduous tree canopies in the Appalachian Mountains. Under these conditions, diffuse light is the major source of light energy in the summer. Additional energy is also received as sun flecks during sunny days. The sun flecks are high pulses of radiation, which occur for a short duration due to gaps in the canopy.
Overall growth of
R. maximum
was better, however, with increased light, which increased flower bud production (2a, 6, and 8).
The Toronto garden is a semi-open site within a treed urban neighborhood. It can be visualized as a 100 by 60-foot opening in an open urban deciduous forest with a few conifers scattered throughout. Very tall deciduous trees are located in the neighbor's garden to the west. These trees shade the garden in the afternoon, from the third week in May until the middle of October. These trees shed their leaves from the middle of October and re-leaf during the middle of May. A conifer screen 20 feet (6 m) in height and still growing is situated on the south boundary. A second screen of 10 feet (3 m) tall conifers is located along the west side of the garden.
Abandoned Christmas trees are placed adjacent to rhododendrons located in the eastern part of the garden from the beginning of January to the beginning of April. This temporary screening serves to block the low-angled south and southwest sun beam from heating leaves during frost episodes.
The majority of the plants are located in the open part of the garden and are not shaded from above. They receive between 2-3 hours of direct overhead sunlight during the summer. The remainder of the day these open-situated plants are shaded from the southwest and west sun beam.
A few plants are located partly under the shade of either a small deciduous tree or coniferous shrubby tree, and receive direct light from the sun beam for one hour from an open side facing the sun. The rest of the day these partially shaded plants receive diffuse-shaded light.
During the fall and winter, the plants receive angled-sunlight; on September 21, the angle is 43 degrees, on December 21, 22 degrees, and on March 21, 43 sun/over horizon angle degrees in Toronto at latitude 44 North (Figure 1).
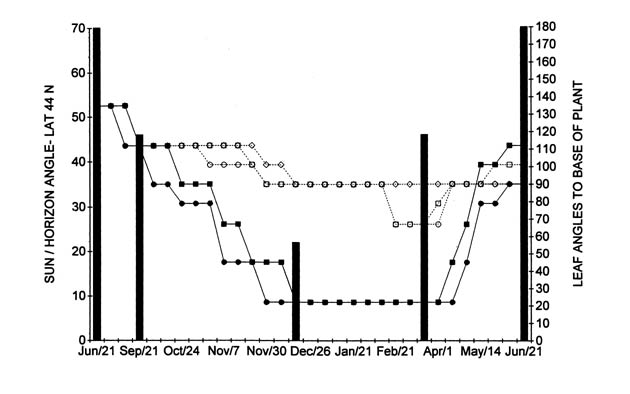
|
|
Figure 1. Current year seasonal rhododendron leaf angles above freezing with
day-length/ light intensity (sun/horizon angle). Hardy (two lower lines ) R. brachycarpum as ssp. tigerstedtii seedling; R. maximum from Tunnel Mountain, VA. |
The Plants
Specimens used for the measurements are mostly registered cultivars (Table I and II). The plants used for leaf angle measurements were purchased from Rhodoland nursery, which is run by ARS members Jack and Jackie Looye in Niagara-on-the-Lake, Ontario. The plants are grown from rooted cuttings and are 3-4 years old with flower buds, when sold in one-gallon pots. They were planted in raised acid beds (pH 5.5-6.0) suitable for the growth of rhododendrons, and had grown between three to eight years in these beds.
Rhododendron maximum
(3 feet, 0.9 m, tall, with late June white flowers) was raised from a "Tunnel Mountain" seed source, in the vicinity of Blacksburg, Virginia, USA.
R. maximum
'Mount Mitchell'
1
(3 feet, 0.9 m tall, with early July light pink flower buds becoming white), and
R. brachycarpum
as ssp.
tigerstedtii
were obtained and grown from the ARS seed exchange.
Measuring Freeze-leaf-curl
A minimum-maximum thermometer is located near the rhododendrons, at 50 cm above ground level and sheltered from the direct sun beam, to obtain an air temperature at the time of measuring. Freeze-Leaf curling is as sensitive to changes in air-freezing
2
temperatures, as the mercury in a thermometer, as long as the measurement of the leaf curl is taken in full shade or semi-darkness. I always measured at dawn when there is enough light to see, but well before the sun could strike the plants to eliminate sun heating and uncurling of the leaves. I also measured during periods of steady freezing temperatures, not during warming or cooling periods.
I selected two leaves per plant from the top shoots (current year's leaves) for repeated leaf-curl measurements throughout the dormant season. I measured residual leaf width with a ruler (cm) at the mid point of the leaf blade (lamina) at temperatures from +1C to 17C. The leaf width measured does not include curl overlap of leaf blade edges, as in (13). The
residual leaf-width measurement
at a particular freezing temperature is expressed as a ratio of
residual leaf width
divided by the
fully extended leaf blade width
. Percent leaf curling = [1-(residual freeze leaf width / leaf width at +1 C air temperature) x 100]. Variation between the measurements of two leaves was less than ten percent for all cultivars and species.
Table I is included to show freeze-leaf-curling for many rhododendron cultivars in Toronto. Most of these plants curled and uncurled their leaves without any observable damage at air temperatures of 12C to 17C, for the dormant season of November 2001 to April 2002. The data does not offer a conclusive difference in leaf curl between cold hardy
R. maximum
(Tunnel Mountain, VA), and very tender R. 'Anah Kruschke'.
Plate I-1, and I-2 illustrate intense freeze-leaf-curling and intense freeze-leaf-angle change for two Hymenanthes rhododendrons,
R. brachycarpum
as ssp.
tigerstedtii
seedling and R .'Calsap'. Plate I-3, and I-4 illustrate the intense freeze-leaf-curl for lepidote rhododendrons PJM Group and 'Windbeam', with only the PJM Group showing irregular freeze-leaf angle change, whereas little or no freeze-leaf-angle change occurs in cold hardy R. 'Windbeam'. Lepidote rhododendrons are excluded from this paper, because of lack of sufficient numbers of these cultivars in the author's garden.
Seasonal Changes in Light Intensity and Duration
Seasonal changes in leaf position, or lack thereof, appeared to the author to be in relation to changes in seasonal light intensity and duration during a calendar year, among other factors. The approach of this exploration evolved simply by taking leaf angle measurements through a calendar year. Using the calendar year as a framework automatically includes the effect of seasonal changes in light intensity and duration, on the leaf-angle measurements.
Seasonal changes in day length and light intensity are caused by the seasonal tilt of the earth north/south axis towards or away from the sun by 23.5 degrees through a calendar year (15). This changes the angle of the solar beam over the horizon from June 21, down to December 21. As a result, in Toronto (also Halifax, Nova Scotia and Eugene, Oregon, USA), located at about 44 degrees latitude north, the sun on June 21 rises in the northeast, moves high across the sky (70 degrees over the horizon at noon), and sets in the northwest, during the longest day in summer, to provide about 16 hours of day light.
During the shortest day on December 21, the sun rises in the southeast, moves low across the sky (22 degrees over the horizon at noon), and sets in the southwest to provide about 8.5 hours of daylight. Light striking horizontal surfaces at a steep 70 degree angle during the Toronto summer spreads out less over a surface than light at a low 22 degree angle in Toronto's winter. The same amount of light energy received by a surface in the summer spreads out over a much larger area in the winter. As a result much less energy is received in the winter (15).
Sun over horizon angles are ten degrees higher for latitude 34 north (Smokey Mountains, the Caucasian mountains, and mountains of Central Japan), than latitude 44 north. At latitudes from 24 to 30 north (the Nepal and southwestern Chinese Rhododendron taxa), sun over horizon angles are 20 degrees higher than 44 latitude north. These are the latitudes where native species of
Rhododendron
subgenus Hymenanthes occur.
Measuring Leaf Angles
One specimen per rhododendron and only current year's leaves were used for the measurements. All leaf angle data presented in tables were obtained with a protractor-like card and a plumb bob (a dead weight on a string) to measure the vertical leaf angles between the leaf blade (lamina) and the base of the plant at ground level. A plumb bob is used to orient the protractor-like measuring card vertically along the Zenith to Nadir axis. Zenith is directly overhead the plant at 180 angle-degrees above ground level (Nadir), which is at zero angle-degrees.
Vertical Leaf Angle Measuring Classes
Angles of four leaves from a leaf-cluster of five to seven leaves were measured to obtain an average leaf angle value for the leaf cluster of each rhododendron cultivar, each represented by one plant. Leaf angle classes were indicated on the measuring card for ranges of 11.5 vertical angle degrees, with 5.625 angles degrees above and below each mid-point of a measuring class. The mid-point of each measuring class was used to show the results in tables, i.e., the mid-point class angle indicated as 90 includes angles between 96 (95.625) and 84 (84.375) on the measuring card.
Measurements were taken from June 21, 2002, to June 21, 2003, and for a second full year thereafter, when the air temperature was above freezing (+2 or more degrees Celsius), as well as during freezing episodes in the dormant season.
| Table II . Current year leaf angles at temperatures above freezing, for selected rhododendrons in Toronto Garden, 2002-2003. | ||||||||||||||||
| Month | June | Sep | Oct | Oct | Nov | Nov | Nov | Dec | Jan | Feb | Mar | Apr | Apr | May | May | June |
| Date | 21 | 21 | 11 | 24 | 7 | 20 | 30 | 20 | 21 | 19 | 18 | 1 | 22 | 3 | 14 | 21 |
| R. brachycarpum as ssp. tigerstedtii | 135 | 112 | 90 | 90 | 45 | 45 | 22 | 22 | 22 | 22 | 22 | 22 | 45 | 45 | 79 | 90 |
| R. 'Calsap' | 135 | 112 | 101 | 90 | 67 | 45 | 45 | 45 | 22 | 22 | 22 | 22 | 67 | 79 | 90 | 90 |
| R. maximum (Tunnel Mt) | 135 | 112 | 112 | 90 | 67 | 45 | 45 | 22 | 22 | 22 | 22 | 22 | 45 | 67 | 101 | 101 |
| R.' Helsinki University | 135 | 112 | 112 | 90 | 67 | 45 | 45 | 45 | 45 | 45 | 45 | 67 | 79 | 90 | 101 | 101 |
| R. 'Haaga' | 135 | 112 | 112 | 90 | 67 | 45 | 45 | 45 | 45 | 45 | 45 | 45 | 79 | 90 | 101 | 101 |
| R. maximum 'Mount Mitchell' | 135 | 112 | 112 | 90 | 79 | 45 | 45 | 45 | 22 | 22 | 22 | 22 | 45 | 67 | 79 | 101 |
| R. 'Catawbiense Album' | 135 | 112 | 112 | 90 | 67 | 45 | 45 | 45 | 45 | 22 | 22 | 22 | 45 | 45 | 67 | 90 |
| R. sutchuenense | 135 | 112 | 112 | 112 | 67 | 67 | 145 | 45 | 45 | 22 | 22 | 45 | 79 | 90 | 90 | 90 |
| R. 'Maximum Roseum' | 135 | 112 | 112 | 112 | 90 | 67 | 67 | 45 | 45 | 45 | 45 | 45 | 79 | 90 | 90 | 90 |
| R. smirnowii | 135 | 112 | 112 | 90 | 79 | 67 | 67 | 67 | 67 | 45 | 45 | 67 | 79 | 90 | 101 | 101 |
| R. 'Saint Michel' | 135 | 112 | 112 | 101 | 79 | 67 | 67 | 67 | 45 | 22 | 22 | 79 | 90 | 101 | 101 | 101 |
| R. 'Nova Zembla' | 135 | 112 | 112 | 112 | 90 | 79 | 67 | 67 | 67 | 45 | 45 | 67 | 67 | 79 | 79 | 101 |
| R. 'Normandy' | 135 | 112 | 112 | 101 | 90 | 79 | 79 | 79 | 79 | 67 | 67 | 67 | 79 | 90 | 101 | 101 |
| R. 'America' | 135 | 112 | 112 | 101 | 101 | 90 | 79 | 79 | 67 | 45 | 45 | 67 | 79 | 90 | 101 | 101 |
| R. 'Looye' unnamed X | 135 | 112 | 112 | 112 | 90 | 90 | 79 | 79 | 67 | 67 | 45 | 67 | 90 | 90 | 90 | 101 |
| R. 'Sumatra' | 135 | 112 | 112 | 112 | 90 | 90 | 90 | 79 | 79 | 67 | 67 | 79 | 79 | 90 | 101 | 101 |
| R. 'Paul C. Bosley' (unreg.) | 135 | 112 | 112 | 112 | 112 | 101 | 101 | 90 | 90 | 90 | 79 | 79 | 90 | 90 | 90 | 101 |
| R. 'Cunningham's White' | 135 | 112 | 112 | 112 | 112 | 101 | 101 | 90 | 90 | 90 | 90 | 90 | 90 | 90 | 90 | 90 |
| R. 'Golden Genie' | 135 | 112 | 112 | 112 | 101 | 101 | 90 | 90 | 90 | 67 | 45 | 79 | 90 | 90 | 90 | 90 |
| Table III . Current year leaf angles at temperatures (C), below freezing, for selected rhododendrons in Toronto Garden, 2002-2003. | |||||||||||||
| Month | Oct | Oct | Nov | Nov | Nov | Dec | Jan | Jan | Jan | Feb | Feb | Mar | Mar |
| Date | 23 | 31 | 16 | 21 | 27 | 3 | 2 | 14 | 22 | 11 | 16 | 3 | 31 |
| Temperature C | -3 | -3 | -7 | -3 | -7 | -17 | -10 | -12 | -21 | -18 | -19 | -24 | -8 |
| R. brachycarpum as ssp. tigerstedtii | 45 | 45 | 45 | 22 | 22 | 11 | 22 | 22 | 11 | 11 | 11 | 5 | 22 |
| R. 'Calsap' | 45 | 45 | 22 | 22 | 11 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| R. maximum (Tunnel Mt) | 45 | 45 | 22 | 22 | 11 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| R.' Helsinki University | 45 | 45 | 22 | 22 | 11 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 11 |
| R. 'Haaga' | 67 | 45 | 22 | 22 | 11 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 22 |
| R. maximum 'Mount Mitchell' | 45 | 45 | 22 | 22 | 11 | 1 | 1 | 1 | 1 | 1 | 1 | 11 | 22 |
| R. 'Catawbiense Album' | 45 | 45 | 22 | 22 | 22 | 11 | 11 | 11 | 1 | 11 | 11 | 1 | 11 |
| R. sutchuenense | 67 | 67 | 45 | 22 | 11 | 11 | 11 | 1 | 1 | 1 | 1 | 11 | 22 |
| R. 'Maximum Roseum' | 45 | 45 | 45 | 22 | 22 | 11 | 22 | 11 | 11 | 11 | 11 | 11 | 22 |
| R. smirnowii | 45 | 45 | 45 | 22 | 22 | 22 | 22 | 11 | 22 | 22 | 11 | 11 | 11 |
| R. 'Saint Michel' | 67 | 67 | 45 | 22 | 22 | 11 | 22 | 22 | 22 | 22 | 11 | 5 | 11 |
| R. 'Nova Zembla' | 67 | 67 | 45 | 45 | 45 | 22 | 22 | 22 | 22 | 22 | 22 | 11 | 22 |
| R. 'Normandy' | 45 | 45 | 22 | 22 | 22 | 11 | 11 | 11 | 11 | 11 | 11 | 11 | 11 |
| R. 'America' | 112 | 112 | 79 | 67 | 67 | 45 | 22 | 22 | 45 | 45 | 45 | 45 | 45 |
| R. 'Looye' unnamed X | 90 | 90 | 79 | 67 | 45 | 45 | 45 | 22 | 22 | 22 | 22 | 11 | 22 |
| R. 'Sumatra' | 101 | 90 | 79 | 79 | 45 | 45 | 45 | 45 | 45 | 45 | 45 | 45 | 45 |
| R. 'Paul C. Bosley' (unreg.) | 90 | 90 | 79 | 79 | 79 | 45 | 45 | 45 | 22 | 45 | 45 | 45 | 45 |
| R. 'Cunningham's White' | 101 | 101 | 90 | 90 | 90 | 79 | 79 | 79 | 79 | 79 | 79 | 79 | 79 |
| R. 'Golden Genie' | 90 | 90 | 90 | 90 | 79 | 67 | 67 | 45 | 45 | 45 | 45 | 45 | 45 |
Results and Their Interpretation
Results of seasonal leaf angle measurements for temperatures above freezing are shown for the period from June 21, 2002, to June 21, 2003, in Table II. The hardy plants are arranged at the top of the Table II and III followed by the intermediate hardy and with the tender one towards the bottom of the table. These results are presented for two hardy (lower two lines) and three tender (top three lines) rhododendrons in Figure 1, together with the sun angle over the horizon for Toronto at about 44 degrees latitude north. The interpretation of the results are presented for four seasonal phases as follows:
1) Initial spring/summer growing season.
All rhododendron plants measured, and many other cultivars in the Toronto garden, initiate their young and current year leaves at a 90+45=135 degree angle position above ground level, from the middle of May to the end of June. The new leaf-clusters develop like miniature shade-umbrellas above last year's cluster of leaves and mature some time after June 21.
Over the summer the leaf angles of the new leaves gradually drift down to a value of 112 angle degrees. Leaves of last year's leaf clusters are held more or less horizontally at about 90 angle degrees, from the beginning of July, below the newly developing leaf clusters.
Some of my rhododendrons, which are situated in shady corners, are exposed on one side of the plant to the full sun beam at some time during the day. They tilt the lower leaves of the leaf cluster down to a 79-degree-angle, and the upper leaves up to a 101-degree-angle. The result is a leaf cluster held in a tilted plane towards the sun beam. The average of these angles is about 90-degrees.
2) The autumn hardening phase.
For most deciduous woody-temperate shrubs and trees, diminishing day length triggers onset of autumn leaf senescence followed by abscission and shedding of leaves. Hymenanthes rhododendrons, in contrast, maintain their leaves in an evergreen condition. Downward movement of leaves of hardy Hymenanthes rhododendrons is a visually noticeable autumn-event that can be captured by measuring changes in leaf angles.
In Table II, and Figure 1, the leaves of several of the rhododendrons in my garden moved downwards at varying degrees during the fall when day and night temperatures remained well above freezing. The most dramatic downward positioning of leaves from 135 to 45 and 22 angle degrees was observed for some of the hardy cultivars, such as
R. maximum
(Tunnel Mountain, VA source), R. 'Calsap', and
R. brachycarpum
as ssp.
tigerstedtii
seedling. Changes in leaf angles for tender ones, such as R. 'Sumatra' and R. 'Cunningham's White' were considerably less, from 135 to about 90, or 79 angle degrees by December 21.
Data presented in Table II, and Figure 1 for the autumn phase, suggest that changes in leaf angle for hardy rhododendrons, when the temperatures are still above freezing, may be related to seasonal changes in day length and/or light intensity, together with decreasing autumn temperatures. For hardy rhododendrons downward movement of leaves in autumn occurs apparently to disengage leaves from possible high light exposure damage during freezing episodes.
On the other hand, tender cultivars, (Figure 1, top lines), show little, or no such leaf angle changes. Cultivars intermediate to the hardy and tender ones, such as R. 'Nova Zembla', show intermediate leaf angle changes with decreasing day length and light intensity.
3) Winter dormant period and freezing episodes.
The results for leaf angle measurements during dormant season freezing periods are shown in Table III.
In the Toronto garden, hardy cultivars which maintain low angles during the dormant season, when the temperature is above freezing, turn down to near zero degrees during freezing periods (Plate I #1 and #2). When the air temperature rises above freezing during mild spells between freezing episodes, the leaves turn up again to their previous downward dormant season 45 degree angle position.
These results are in agreement with Nilsen, (5, 11, and 12) who noted that further downward movement of leaves, can be triggered by freezing temperatures during the dormant season, apparently to minimize high light damage impact on the photosynthetic pigments.
Cultivars that maintain near horizontal leaf positions in the dormant season (Table III) show much less downward movement caused by freezing temperatures and may temporarily turn down their leaves to 79 degrees. These cultivars tend to be the marginally hardy ones in Toronto, i.e., R. 'Cunningham's White', and R. 'Sumatra'. The leaves of these two cultivars were partially damaged by the freezing temperature of 24C, which occurred on March 3, 2003. Rhododendron 'Golden Genie' obtained from Vancouver, Canada, was severely damaged by the March 3, 2003, temperature of 24C. It lost all its last year's leaves and then formed new leaves in early May and promptly died shortly after.
Different leaf angle values above freezing were obtained for some of the rhododendrons, when comparing angle values for different years. Freezing episodes (Table III) occurred regularly throughout the dormant season of 2002-2003. The winter of 2002-2003 was colder than a normal Toronto winter, and as a result the rooting zone remained frozen for at least one foot down and well into April.
The winter of 2003-2004 was a normal cold Toronto winter, with a relatively mild December and dormant season with a one and a half-foot deep snow cover until the beginning of March, followed by a freezing episode during the middle of March. As a result the soil was not as deeply frozen and thawed out by the beginning of April. Leaf angle values for
R. maximum
and others were at higher leaf angle values (45) as compared to the previous winter season (22).
These less down-turned leaf angle values for the 2003-2004 winter as compared to the winter before may indicate a less hardened-off state for the plants with a greater chance of frost damage. Many Toronto-Region members reported extensive flower bud damage for this 2003-2004 winter season, whereas no such damage was apparent for the 2002-2003 colder than normal Toronto winter.
4) Spring day length effect on leaf angle changes.
A combination of increasing day length, light intensity and temperatures suitable for growth triggers the upward movement of leaves, by April followed by leaf bud break. Last year's leaves move from lower leaf angle positions to 90 degrees (horizontal) and for short periods up to 101,112 and then down to 90 degrees by July 1, when the current year's leaves completed the development of the leaf laminas.
The current year's new developing leaf clusters act like miniature umbrellas over last year's leaf clusters and begin to shade them. It may be that in response last year's leaf clusters extend the leaves about horizontally for optimum light reception. The upward movement of leaves is much less pronounced in tender plants, because leaf angles do not turn down in autumn as much.
An educational flower forcing display illustrates the effect of artificially increasing day length, light intensity and temperatures suitable for growth, which triggered the upward movement of leaves. This display with tender and reasonably hardy rhododendrons was placed near the south facing glass roof-wall in the Toronto Civic Garden Centre. Leaves were turned from 79 downward to 45 degrees during the last week of February, depending on the cultivar. A bank of growth light provided sixteen (Toronto, June 21) summer-day-length hours in addition to the natural light coming in through the glass roof-wall. At the time (March 31) when the flower buds flushed inside the building, all the last year's leaves had turned up to 90-101 degree leaf angles, apparently in the more advantageous position to intercept light for photosynthesis.
Light intensity from the end of February until March 31st was extended by overhead grow-lights to simulate outside mid-summer length (16 hrs). This amount was still considerably less than the light intensity that would have been received for the third week in May to June 21st. The increase in day length to 16 hours for the educational display together with room temperatures, apparently were the most likely factors that triggered the upward leaf movement in preparation for full photosynthesis, followed by flower bud break. This collection of hardy and tender specimen was planted outside in Edwards Gardens, Toronto, after the show. These cultivars normally would only flower between the third-week of May till the end of June in Toronto.
Relative Light-intensity Percentages
The focus of this leaf-angle-exploration project is on seasonal light intensity and its possible effects on leaf angle changes. Light intensity is of interest because of its direct bearing on photosynthesis, photoinhibition and the capability of hardy rhododendrons to position leaves at different angles seasonally to deal with the effect of incoming light during freezing episodes. A calculation, therefore, is presented to estimate potential light percentages reaching leaf clusters of rhododendrons as follows:
Light intensity: The sum total of light energy reaching any cluster of leaves not only varies throughout the day but also varies through the seasons, due to seasonal changes in day length and light intensity. The relative amount of light received in Toronto at 44 degrees latitude north can be calculated as a percentage of light received at the equator for June 21, September 21, December 21, and so on, using an arbitrarily chosen-8-inch diameter circular leaf cluster area, as follows: The maximum amount of light (100 percent) received from the sun beam standing overhead at the equator at noon (90 degrees sun / over horizon angle) is taken as the reference point for the circular area of 8 inches in diameter with leaves held horizontally.
At latitude 44 north, the sun beam at noon stands at 70 angle degrees over horizon on June 21. The maximum amount of light received at 44 degrees latitude north is 81 percent from the amount at the equator. On September 21 and March 21 when the sun angle is 46 degrees over the horizon at noon, the amount of light reaching the circular area is about 42 percent.
On December 21, when the sun angle is reduced to 22 degrees over the horizon, the light received is about 18 percent.
Effect of Leaf Angle Changes on Light Reception
Hardy rhododendrons as indicated in the previous sections adjust their leaf position to regulate the amount of energy striking the leaves. Combining seasonal light percentage reception at latitude 44 north, with leaf angle reduction percentages would provide a relative percentage-estimate of light received by rhododendrons through the seasons. This is calculated as follows: The amount of light received by a cluster of leaves forming a circular area of any diameter can be used to explore the effect on light reception by moving leaves upward or downward. For example a cluster of 4 inch (10 cm) long leaves, held horizontally (90 degree angle) forming a circular area of 8 inches in diameter (B)=(50 square inches), is used for the calculations. The largest circular exposure to light (100 percent) can be obtained by extending the leaves horizontally (90 degrees).
The 8-inch diameter (B), of the circular area at 135 and 45 leaf angle degrees is reduced to 5.8 inches (A), and the circle area receiving light = about 26.4 square inches. This reduces light reception to 53% (26.4/50) from the circular area of 8 inches in diameter with leaves held horizontally.
At an angle of 22 above the base of the plant the diameter of the circular area receiving light is reduced to 3.2 inches (8 cm), with a circular are of about 8 square inches. The light received by the 3.2-inch diameter area is 16% (8/50).
At 11 degrees above ground the percentage is about 4 and zero when the leaves are vertically down. Combining seasonal light percentages with leaf angle reduction percentages would provide a relative percentage-estimate of light received through the seasons, as shown in Table IV. Other factors that modify light reception are discussed under the Discussion heading "Light Reception." Table IV can be used to illustrate light-sensitivity response through leaf angle change. Such inherent responses are marked for hardy
R. brachycarpum
as ssp.
tigerstedtii
and tender R. 'Cunningham's White'.
My research indicates that hardy rhododendrons indeed turn down their leaves to receive minimal light exposure to prevent photoinhibition at above freezing temperatures and freeze thaw damage during freezing periods. In contrast, tender rhododendrons maintain more or less horizontal leaf positions above freezing temperatures. Tender rhododendrons furthermore appear to have little or no ability to move leaves downward during freezing episodes.
One can use the leaf angle values in Table II and III to determine the potential light perception patterns for all rhododendrons listed. These would show patterns that fall within those shown for hardy
R. brachycarpum
as ssp.
tigerstedtii
and tender R. 'Cunningham's White'.
| Table IV. Seasonal percent light reception at latitude 44 north by circular areas held at varying angle-degrees. The chart also shows relative light reception percentage amounts, due to changes in angles for R. bracycarpum as ssp. tigerstedtii (@), and R. 'Cunningham's White' (#). Note: row #2 = (x%); column #2 = (x%). Light percentage values in the chart are obtained by multiplying percentages from row #2 with percentages from column #2. | ||||||||||||
| Leaf | June | Sept. | Oct. | Nov. | Dec. | Jan. | Feb. | Mar. | Apr. | May | June | |
| angle | 21 | 21 | 21 | 21 | 21 | 21 | 21 | 21 | 21 | 21 | 21 | |
| (X%) | (81) | (43) | (35) | (27) | (18) | (27) | (35) | (43) | (56) | (69) | (81) | |
| 135 | (53) | 43@# | 22 | 19 | 14 | 10 | 14 | 19 | 22 | 30 | 37 | 43 |
| 112 | (81) | 66 | 35@# | 28 | 22 | 15 | 22 | 28 | 35 | 45 | 56 | 66 |
| 101 | (90) | 73 | 38 | 32 | 24 | 16 | 24 | 32 | 38 | 50 | 62 | 73 |
| 90 | (100) | 81 | 43 | 35# | 27# | 18 | 27 | 35 | 43 | 56# | 69@# | 81@# |
| 79 | (90) | 73 | 38 | 32 | 24 | 16# | 24# | 32# | 38# | 50 | 62 | 73 |
| 67 | (81) | 66 | 35 | 28@ | 22 | 15 | 22 | 28 | 35 | 45 | 56 | 66 |
| 45 | (53) | 43 | 22 | 19 | 14@ | 10 | 14 | 19 | 22 | 30 | 37 | 43 |
| 22 | (16) | 13 | 7 | 6 | 4 | 3 | 4 | 6 | 7@ | 9@ | 11 | 13 |
| 11 | (4) | 3 | 2 | 1 | 1 | 1@ | 1@ | 1@ | 2 | 2 | 3 | 3 |
| 0 | (0) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Discussion and Summary
Because large-leaf evergreen rhododendrons retain their leaves through the dormant season, some of these species developed leaf movement mechanisms to counter photoinhibition and frost damage of the leaves in contrast to another group of rhododendrons, the deciduous azaleas.
Leaf movements: hardy vs. tender
. Leaf angle movements of hardy rhododendrons of subgenus Hymenanthes correlate with seasonally changing light intensity and day length (Table II, Figure 1), and freezing temperatures (Table III), among other contributing factors such as leaf water stress and overhead canopy effects on light.
Tender cultivars in Toronto respond to diminishing light duration and intensity in autumn, by maintaining leaves in a more or less horizontal plane. This would provide the best leaf position for light reception in the fall and dormant season. This also creates the greatest possibility for damage during freezing episodes in cold climates. Tender rhododendrons likely continue photosynthesis during the winter months in more southern locations. Fewer freezing periods would occur, and leaf temperatures can be more easily maintained at 10 degrees Celsius or more (8).
There are a number of aspects involved in the leaf angle changes or lack thereof:
a) A seasonal non-freezing response
. The seasonal non-freezing one causes downward movement of leaves of hardy
Rhododendron
in the autumn, apparently to disengage the leaf from excessive light-exposure that can cause photoinhibition, and freeze-thaw-injury in the dormant season.
Two environmental hardening phases may be involved to trigger downward movement of rhododendron leaves in autumn. After shoot growth is completed, a first stage of autumn cold hardening is initiated at a critical day length, specific to cultivars and species, when the temperatures are above 5C (41F). A second stage of hardening is induced by temperatures between 0 to 5C (32-41F), as outlined in the review by Vainola (16).
Genes of hardy rhododendrons, furthermore, respond to the environmental autumn hardening phases through control of succulence of leaves and low water stress/elasticity factors that are involved in leaf angle and leaf curling events (13).
The spring-upward movement of leaves re-engages the leaf to receive more light. There is an upward movement to 90, 101 and 112 angle degrees depending on the species or cultivar, and then downward to 90 degrees as the new leaf clusters forming above start shading last year's leaf clusters below. The leaf angle for the new current year's leaf clusters is maintained through the summer at 135 angle degrees but gradually declines to 112 by September 21. It may be that the juvenile leaves need to reduce light intensity by maintaining this 135 degree angle rather than a 90 degree angle. It may be that the 135 degree angle also serves as a means to allow more light to reach last year's whorl of leaves below, which are held at 90 degrees from June 21, throughout the summer.
In northern continental climates, hard freezes (-10 to-20C) may occur in some years during this spring period and may damage hardy as well as tender cultivars. Furthermore this upward turning of leaves that increases susceptibility to mid- and late-spring frosts, is proving to be a barrier in growing rhododendron cultivars much further north than the Toronto Region.
b) A freezing temperature response
. The freeze-temperature angle response (Table III) occurs simultaneously with the freeze-leaf-curl response (Table I). freeze-leaf-curl also provides for leaf surface reduction. Together with down turning of leaves, a considerable reduction of the leaf surface is accomplished in order to receive minimal light exposures, depending on the species or cultivar, during dormant season freezing episodes.
c) The "Herb Spady dormant season effect."
Herbert Spady (14) observed in 1994 that frost damage to flower bud, leaf or plant can occur through a range of temperatures. Different states of hardening during a dormant season can be responsible for frost damage through a range of temperatures. Different leaf angle values measured for different dormant seasons may also indicate different levels of hardening with the possibility of damages occurring through a range of freezing temperatures.
d) Impact of water stress on leaf angles
. Low winter leaf angle positions are also affected by leaf-water potential, especially when soils are frozen. Leaf water potential is lower in the winter than the summer, noted Erik Nilsen in a written communication: "Leaf-water potential is determined by a number of factors among others:
1) Soil water availability and soil temperature; low soil temperature in the winter decreases soil water potential and may reduce water uptake by roots.
2) In the winter leaf vapor pressure is lower in the atmosphere and stems may have suffered some embolism, which reduces water movement through the stems (hydraulic conductance). Embolism of stems may be caused by dehydration in the winter (winter cavitation), which leads to the expansion of gas bubbles in the xylem, and which may cause injury to the ray-protoplasm of the xylem" (3).
Furthermore, small amounts of moisture transpire through the outer skin (cuticle) of the leaves when stomata are closed at leaf and air temperatures below 10 degrees Celsius (8). Over a period of several winter months, especially when the rooting zone is frozen, leaves can dehydrate without moisture replenishment coming from the roots. This situation is amplified when the plants are exposed to frequent strong winds. This affects the water potential in the leaves and may contribute to the downward movement of leaves, both during episodes when temperatures are either above or below freezing.
Light Reception
. Calculated relative light reception percentage data was presented under Results and Their Interpretation. As compared to the equator, plants at latitude 44 north would receive on June 21, 81% of the light. On September 21, the amount is 43%, on December 21, 18%, and then rises for March 21 to 43% and June 21 to 81%. There are some factors that may modify the actual amount and quality of light reaching the plants seasonally.
Canopy Effects on Light Reception
. In a written communication, Erik Nilsen noted that only a portion of the light impinging on a leaf is derived directly from the solar beam. A large portion of radiation arrives as diffuse radiation rather than directly from the solar beam. Diffuse radiation is reflected off clouds and other surfaces, and it increases as the solar angle decreases going into the dormant season, due to the longer path length of the solar rays through the atmosphere. In addition the amount of radiation in the winter can be larger or smaller than that of the summer depending on the nature of the canopy. Under deciduous tree canopies the light can be higher in the winter than the summer (2a).
Another effect of partial shading is the summer tilting of leaf clusters towards the incoming sun beam that shines through an opening in the canopy.
In milder regions to the south of the Great Lakes rhododendrons usually grow in gardens, or forests with shaded, dappled-light situations below deciduous tree canopies with some conifers. Such a canopy shades the rhododendrons during the summer. An excellent example of a mainly deciduous tree canopy with dappled light is shown in the illustration of the Zurich Garden, Toms River, New Jersey, on page 183 of the 2003 fall edition, of the Journal of ARS.
Tree Canopy and Winter Sun Shelter
. In our continental Toronto climate we can hardly grow rhododendron cultivars and species below closed canopies of deciduous trees, because the sunlight would be blocked out during the summer. This is the main period when photosynthesis can take place in the north. Furthermore, the open nature of deciduous tree canopies during the winter, often cause photoinhibition damage, and or freeze/thaw injury (3).
Evergreen rhododendrons grown in continental climates, as in Ontario, must therefore be provided with shelter from direct exposure to the winter sun beam. In Toronto gardens without closed deciduous or coniferous canopies, permanent conifer screens, fences, hedges, and abandoned Christmas trees are used to block the incoming low-angled winter sun beam from the southwest, south and southeast side of the garden. This is the "b" winter sun shelter situation (4). Winter sun shelter of the evergreen plants remains a must in the Toronto Region, regardless of how hardy the cultivars are.
Cold Stress vs. Water Stress Tolerance
. Nilsen raised the question whether cold stress tolerance is coupled with water stress tolerance. Nilsen noted that "if winter hardiness is significantly impacted by leaf movement and leaf movement require succulent leaves, with low water stress tolerance, then there may be a trade off between winter hardiness and water stress tolerance for cold hardy and tender rhododendrons" (13).
High water stress tolerance in less hardy and non-hardy rhododendrons would be a desirable trait for cultivars and species that grow in southern climates with higher temperatures, longer seasons and longer water stress periods. For cold hardy plants the opposite trait of low-water-stress tolerance would be desirable in order that leaves may curl and change leaf angles during cold-stress periods.
Data on leaf-angle changes in this paper is in agreement with the research by Nilsen and others, and re-affirms that the hardier rhododendrons move their leaves down during the dormant season to reduce the amount of damaging light striking the leaves. Cultivars or species without significant leaf movements would receive larger doses of damaging light during freezing periods, which would affect their survival during the winter period from December to May.
The practical task ahead would be to take seasonal leaf angle measurements above freezing for all new introductions, from June 21 to January 21. During this measuring period, cold hardy rhododendron would decrease their leaf angle to 45, or 22 and during freezing episodes to zero angles. Water stress tolerant but cold-tender rhododendron would show little or no change from the 90 angle degrees downward.
Notes:
1
This selection comes from Mount Mitchell, a 6700-foot mountain, along the southern part of the Blue Ridge Parkway in South Carolina, USA
2
I measure air temperatures, because I neither have capability nor skills to measure leaf temperatures.
References
1. Bao, Y. and Nilsen, E.T. 1988. The ecophysiological significance of leaf movements in
Rhododendron maximum
. Ecology 69:1578-87.
2a. Lipscomb, M.V. and Nilsen, E.T. 1990. Environmental and physiological factors influencing the natural distribution of evergreen and deciduous ericaceous shrubs on northeast and southwest slopes of the southern Appalachian mountains. I. Irradiance tolerance.
American J. of Botany
77:108-115.
2b. Lipscomb, M.V. and Nilsen, E.T. 1990. Environmental and physiological factors influencing the natural distribution of evergreen and deciduous ericaceous shrubs on northeast and southwest slopes of the southern Appalachian mountains. II. Water relations.
American J. of Botany
77:517-526.
3. Lipp, CC and Nilsen, E.T. 1997. The impact of sub-canopy light environment on the hydraulic vulnerability of
Rhododendron maximum
to freeze-thaw cycles and drought.
Plant, Cell and Environment
20:1264-1272.
4. Morsink, W.A.G., and R. Dionne, 2002. Rhododendron Hardiness along the Northern Edge: The Ontario-Quebec Situation.
J. Amer. Rhod. Soc.
56:188-194.
5. Nilsen, E.T, 1985, Seasonal and diurnal leaf movements of
Rhododendron maximum
L. in contrasting irradiance environments.
Oecologia
65:296-302.
6. Nilsen, E.T. 1986. Quantitative phenology and leaf survivorship of
Rhododendron maximum
in contrasting irradiance environments of the southern Appalachian mountains.
American J. Botany
73:822-831. 7. Nilsen, E.T. 1987. Influence of water relations and temperature on leaf movements of
Rhododendron
species.
Plant Physiology
83:607-612.
8. Nilsen, E.T., and Y. Bao 1987. The influence of leaf age, season and microclimate on the photosynthetic apparatus of
R. maximum
. 1. Chlorophylls.
Photosynthetica
21: 535-542.
9. Nilsen, E.T., D.A. Stetler, and C. Grassman, 1988. The influence of age and microclimate on the photo-chemistry of
Rhododendron maximum
L. leaves. ll. Chloroplast structure and photoynthetic light response.
American J. of Botany
75:1526-1534.
10. Nilsen, E.T. 1990. Why do Rhododendron leaves curl?
Arnoldia
50:30-35.
11. Nilsen, E.T. 1991. The relationship between freezing tolerance and thermotropic leaf movements in five Rhododendron species.
Oecologia
87:63-71.
12. Nilsen, E.T., 1992. Thermostatic leaf movements: a synthesis of research with Rhododendron.
Bot. J. Linnean Soc.
110:205-233.
13. Nilsen, E.T., 1993. Does winter leaf curling confer cold stress tolerance in Rhododendron?
J. Amer. Rhod. Soc.
47:98-104.
14. Spady, H.A.1994. Hardiness redefined.
Amer. Rhod. Soc.
48:65.
15. Seeds, M. A, 2004.
Horizons: Exploring the Universe
, Publisher: Brooks/Cole-Thomson Learning, Belmont CA USA. 8th Edition.
16. Vainola, A., 2000. Genetic and physiological aspects of cold hardiness in Rhododendron. Academic dissertation in Plant Breeding. Dept. of Plant Physiol., U. Of Helsinki, Finland. Electronic publication at http://ethesis.helsinki.fi/.
Willem Morsink is a member of of the RSC Toronto Chapter - ARS District 12.